Prof. dr. M. (Martijn) Rep
- Science Park 904
- Room number: C2.218
-
Postbus 1210
1000 BE Amsterdam
-
Research
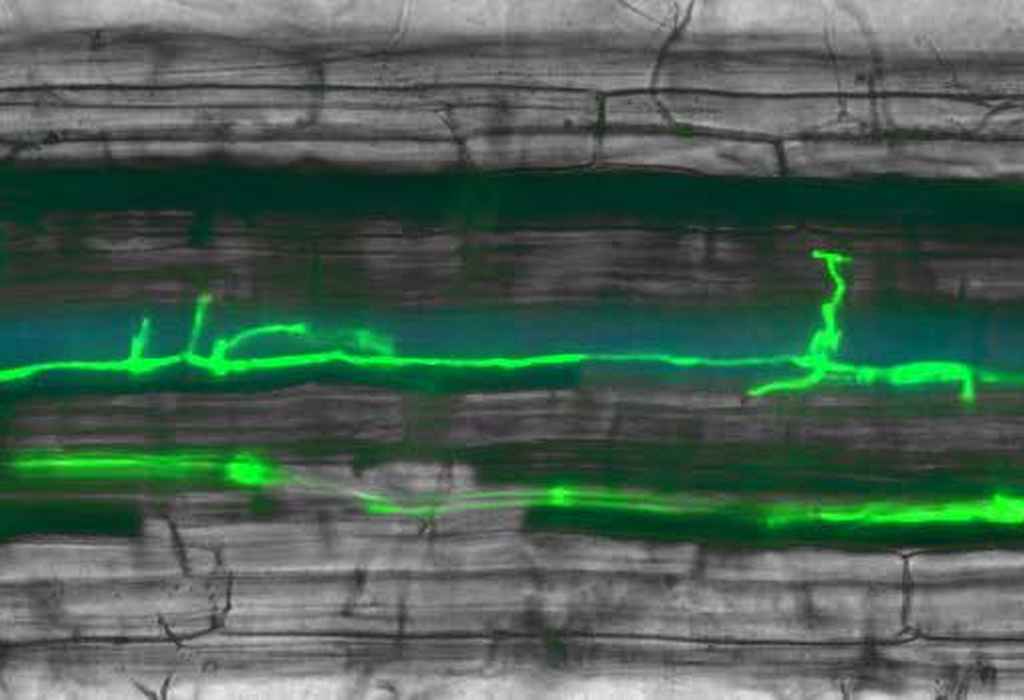
Fusarium oxysporum in a tomato root cell. Fusarium is green fluorescent during invasive growth because it expresses GFP driven by the SIX1 promoter. Since 2000, my group aims to uncover the molecular basis of pathogenicity of the plant-pathogenic fungus Fusarium oxysporum, a fungus that causes wilt disease or root/bulb-rot in many plant species of agricultural and horticultural importance.
Current research includes the identification and charaterization of genes required for pathogenicity, especially those that encode effectors : proteins secreted during colonization of host plants. These effectors are often required for full virulence towards a specific host plant. Some, however, are recognized in resistant plants carrying a matching Resistance gene , in which case they are (also) referred to as avirulence factors. Expression of effector genes is highly upregulated during plant (root) colonization, and we are studying the molecular mechanisms underlying this induction of expression.
In F. oxysporum, effector genes reside in pathogenicity chromosomes. These chromosomes are transposon-rich, can transfer between clonal lines and appear to evolve faster than the 'core' genome. This discovery led us to study molecular mechanisms underlying genome evolution in relation to host-specific pathogenicity.Expedition to investigate Fusarium wilt of banana in Java – November 2016 (KNAW SPIN project led by Prof. Gert Kema) -
Research team
Group members
Babette Vlieger, PhD student; host-jumping by Fusarium oxysporum in cucurbits [EPS-TKI]
Slavica Janevska, post-doc; Role of histone modifications in gene expression, genome stability and pathogenicity in Fusarium oxysporum [personal DFG grant]
Hannah McDermott, technician; Fusarium oxysporum f.sp. tulipae ("Nieuwe middelen voor beheersing van Fusarium ziekte in tulp")' [TKI]
Like Fokkens, post-doc, bioinformatician; Role of transposons in genome evolution in Fusarium oxysporum; [NWO-Veni]
Petra Houterman , technician; Molecular genetics of Fusarium oxysporum; [UvA-SILS]
Previous group members
Maria Constantin, PhD student; Mechanisms and genes involved in biocontrol capacity of Fusarium oxysporum; [ITN BestPass]
Jiming Li, PhD student; Analysis of pathogenicity chromosomes of Fusarium oxysporum; [CSC scholarship]
Sri Widinugraheni, PhD student; Identification and characterization of effectors of F. oxysporum f.sp. cubense; [KNAW-SPIN]
Mara de Sain, technician; Markers for formae speciales of Fusarium oxysporum; [EFRO]
Peter van Dam , PhD student; Identification and characterization of effectors of F. oxysporum pathogenic to various host plants; [NGI Zenith]
Lotje van der Does, post-doc; Communication between core and accessory genomes of F. oxysporum; [NWO-Veni]
Nico Tintor, post-doc; Effector entry into plant cells; [NWO-Vici]
Like Fokkens, post-doc, bioinformatician; Evolution of the accessory genome of F. oxysporum; [NGI-Zenith]
Mara de Sain, PhD student; Targets of effectors of F. oxysporum in tomato; [STW]
Sarah Schmidt, post-doc; Comparative genomics of F. oxysporum; [NWO-Vici]
Ido Vlaardingerbroek, PhD student; Chromosome transfer in F. oxysporum; [NWO-Vici]
Shermineh Shahi, PhD student; Chromosome transfer in F. oxysporum; [NWO-Vici]
Patrick Mak , technician; Proteomic analysis of phloem sap; [TTI Green Genetics]
Vladimir Krasikov , Post-doc; Functional analysis of the tomato xylem sap protein XSP10
Caroline Michielse, Post-doc; Insertional mutagenesis of F. oxysporum, pathogenicity genes; [Utopa foundation]
Linda Reijnen , technician; Insertional mutagenesis of F. oxysporum, pathogenicity genes
Wilfried Jonkers , PhD student; Analysis of FRP1, metabolic requirements of root invasion
Lotje van der Does, PhD student; Analysis of the effector and avirulence factor Six1, horizontal chromosome transfer
Ringo van Wijk , technician; Insertional mutagenesis of F.oxysporum; [Utopa Foundation]
Michiel Meijer, postdoc; Molecular Genetics of F. oxysporum
Other collaborators within Molecular Plant Pathology at the UvA
Prof. Ben J.C. Cornelissen , former head of Molecular Plant Pathology
Dr. Frank Takken and his team members (see link below)
Dr. Harrold van den Burg and his team members (see link below) -
Biography
After graduating in molecular biology at the University of Amsterdam in 1991, I worked as a PhD student in the lab of Molecular Biology headed by Prof. Leslie Grivell. In 1996, I defended my thesis on the role of proteases in mitochondrial biogenesis in the yeast Saccharomyces cerevisiae .
I then moved to Leuven, Belgium, where I worked as a postdoc in the lab of Prof. Johan Thevelein on the response of yeast to osmotic stress, primarily dealing with regulation of transcription.
In 1999, I came back to Amsterdamto work for a year in the lab of Prof. Henk Tabak, setting up microarray technology to study yeast's transcriptional response to fatty acids.
In 2000, I became assistant professor in the Plant Pathology group of Prof. Ben Cornelissen.
From 2002 to 2007, I was research fellow of the Royal Dutch Society for Arts and Sciences.
In 2010 I obtained a NWO-Vici grant to study chromosome transfer in fungi.
In 2018 I became full professor and chair of Molecular Plant Pathology
-
Adaptation, Innovation and Progress
Adaptation, Innovation and Progress - inaugural lecture delivered on 11 April 2019
Introduction
Evolution is the great paradigm of biology; “nothing in biology makes sense except in the light of evolution”, is the title of an essay written in 1973 by the biologist Dobzhansky[1]. The paradigm of evolution is the great explainer in biology. When we ask, for instance: Why do rhino’s have horns? Why do some ants grow fungus gardens? Why do birds have feathers? And a million other ‘why’ questions, the universal answer from the evolution paradigm is: because these things have increased the average number of fertile offspring. In other words, they enhance the fitness of the organism. It is the universal answer to the ‘why’ question. But we do not usually know the answer to the ‘how’ question. Do we know, for instance, how the horn of a rhino enhances its average number of offspring? This is where our imagination is triggered: maybe because of x, presumably because of y, likely because of z. This is much more difficult: what could be the particular benefit of this particular feature of this particular organism? Yet, the underlying assumption is that it must have some benefit, some contribution to the average number of offspring, otherwise it could not exist – it could not have survived the relentless operation of selection of the ‘fittest’ or even the slower but equally relentless process of accidental loss over multiple generations.
Another essential aspect of the paradigm of evolution is that life is not the same as it was before. There was a time, for instance, when there were no living cells on this planet. There was a time when there was no photosynthesis – no cells that produce oxygen by splitting water with energy from sunlight. There was a time when there was no life on land, no birds, no mammals. There was a time when there were no flowers. All life has the same DNA and the same genetic code, and is therefore highly likely to have evolved from the same ancestral cell, the invisibly small mother of all cells. From this microscopic mother cell, over billions of years a stunning diversity of life forms has evolved. Most of the ‘branches’ of this immense tree of life have died out through catastrophic events, such as the impact of meteorites. The present day endpoints of all branches of the tree of life, that we call species, are all super-survivors. All current species have the same evolutionary age and all are equally successful. There really are no ‘old’ or ‘young’ species, terms that are sometimes used by biologists, which I think is somewhat confusing.
In any case, when we compare the life forms around us, including ourselves, with the tiny and simple ancestral mother cell, we cannot deny that something wonderful, something amazing has happened. Surely there has been progress! Tremendous diversification has happened. A tremendous increase in complexity has happened. Tremendous innovation has happened in these billions of years on this planet. Surely, we can confidently say that life has made progress. Or can we?
I will return to this question later. But let us first look at another evolutionary process, the evolution of science – the uniquely human enterprise through which knowledge and technology evolve. A similar story seems to unfold as has happened in biological evolution. In human history, for a very long period knowledge and technology appears to have evolved very slowly, until a few centuries ago when acceleration set in. Whatever the causes of that acceleration, we now are used to hear almost daily of scientific progress in terms of new theories, new discoveries and new technology. But again, for a long historical time we can hardly see progress – mostly a succession of equally valid or invalid theories, just various permutations and adaptations of existing technology. But when we look carefully we do recognize some real innovations in technology: the use of stone tools, the control of fire, the use of metals, the emergence of agriculture and the domestication of animals, to name a few. In such cases, we say that human civilization has advanced from a more primitive to a more sophisticated state. Civilization advanced both in knowledge – of how the material universe works – and in applying that new understanding through technology. However, do advances in understanding and technology always constitute progress? What if this new understanding and new technology is used in a way that undermines human dignity or leads to extinction of species? Can we really call that progress?
For today, I decided to take on the challenge to see to what extent the emerging understanding of the mechanisms underlying the process of biological evolution can be used to inform our understanding of the most pressing questions concerning the advancement of human civilization and the sustainability of the diversity of life on this planet. I will do so by exploring three concepts: adaptation, innovation and progress. For each of these three concepts I will try to see how insights from biology may be applied to science, as well as to society at large.
Adaptation
So let’s first look at the concept adaptation. Every living organism adapts to its environment, by which we mean that it changes its internal state and the way it interacts with its environment to improve either its own survival or the survival of its offspring or its kin. Adaptation happens on two different levels. On one level, adaptation is physiological – it does not involve changes in its DNA. Instead, gene expression changes, growth patterns change, metabolism changes. All organisms do this, from tiny bacteria to giant trees. On another level, adaptation is genetic. On this level, it is actually not an individual that adapts, but a population. This is the process that drives biological evolution. It involves changes in the DNA that are passed on to the offspring. The advantage of this type of adaptation is that it is heritable, and thereby durable: an entire population becomes ‘adapted’ to a new environment. However, changes in DNA are risky, because the vast majority of spontaneous changes in DNA are detrimental to survival or to the average number of viable offspring. All organisms therefore have very intricate cellular machineries to prevent changes in DNA at all costs. Nevertheless, no system is perfect, changes in DNA do happen and, therefore, genetic adaptation is possible and evolution happens.
In the field of plant pathology – which is the study of plant diseases caused by microbes – we have seen many examples of adaptation of pathogenic fungi, bacteria and viruses to a resistant variety of a crop that is introduced in the field. Adaptation in this case means the overcoming of resistance in the crop, allowing the proliferation of the pathogen. We have learned, also in my research, that genetic changes underlying this adaptation can be mutation of individual genes and sometimes even the gain of genes from another species through so-called ‘horizontal transfer’.
In the enterprise that we call science, I propose that adaptation also occurs on two levels, in analogy to the physiological and genetic levels in biology. Any human enterprise takes place in a particular environment or system. The immediate environment for the scientific enterprise is the university. A university provides the conditions for the generation of new knowledge by experimentation, observation and reflection. It provides fuel for this process in the form of money, infrastructure and administration. It facilitates access to knowledge and the preservation of knowledge. And it facilitates the training of students for the continuation of the enterprise. Universities, in turn, are part of a larger environment or system that includes governments, granting agencies and science publishers. An individual scientist can be said to adapt to this environment when she or he enters it and then further adapts when the environment changes. For instance, she will adapt her grant proposals to new guidelines of a granting agency. She will adapt her publication strategy to the incentives for open access publication. She will adapt the content of the course she teaches to the curriculum and adapt the way she grades students to the rules of examination. She may also adapt to changes in how scientific performance is measured, and so on.
All these adaptations can be seen as ‘physiological’: they do not constitute a fundamental, heritable change in behaviour but are opportunistic, aimed to improve ‘survival’ or ‘success’ as defined by the system that the scientist is part of. I suggest that on the level of human enterprises such as science, there is also a process analogous to genetic adaptation, to changes in DNA in populations. This process concerns changes in what I call mental models[2]. Changes in our mental models translate to changes in our behaviour and are heritable, meaning they can be passed on to the next generation. And they can spread in the same generation through ‘horizontal transfer’. Take, for example, collaboration of a scientist with a company. This collaboration can be ‘physiological’ or ‘opportunistic’ when he simply needs it to obtain a research grant that requires a partner from industry. It is survival-focussed. A ‘genetic’ adaptation, on the other hand, is a change in the mental model of the scientist from something like “collaboration with industry is a good thing when it increases my output or strengthens my position in some other way” to something like “collaboration with industry is a good thing for the betterment of society, regardless of my own benefit”. So, mental models have to do with convictions. They lead to a certain behaviour independent of immediate ‘survival’ or ‘success’ in terms defined by the system. I propose that mental models are the DNA of the human spirit.
So now, how could these ideas around the concept of adaptation be applied to the betterment of society, or at least to the preservation of the health of the planet?
Let’s first look at sustainable agriculture. We know that pathogenic microorganisms, insect pests and other enemies of plants will not go away and, unfortunately, will adapt to any changes we introduce in terms of genetic resistance in crops or plant protection chemicals. Of course, plants would not be around today if they did not also adapt to evolving pathogens and pests. For instance, plants can adapt to pathogens through genetic changes in components of their immune system called immune receptors. These immune receptors can recognize invading microbes. The resulting process of mutual adaptation of pathogens and plants has been going on for millions of years and is sometimes described as an arms race or as molecular warfare. However, something has changed around 10.000 years ago, when humans started to domesticate plants to become crops. Crop plants typically have much less genetic diversity than their wild ancestors and are therefore much more limited in possibilities for genetic adaption. To sustain our crops, we have to step in and help them adapt. We can do this, again, at two levels. We can help them adapt physiologically, by enhancing their resilience through application of, for instance, beneficial microbes or through soil improvement. We can also do it genetically, for instance by introducing immune receptors from wild relatives of crop plants through breeding, or, to take it a step further, to make directed changes in plant immune receptors based on our understanding of how these receptors work – in other words, by ‘intelligent design’.
At the level of mental models, intelligent design is of course already being used. Changes in our mental models, in our convictions, are being consciously introduced to suit a particular purpose. Some call it by negative terms such as manipulation or indoctrination. Alternatively, this activity of consciously building mental models is known by more positive terms such as education. It seems to me that the choice of negative or positive terms depends on what we believe the intention is of those that are trying to change our minds. The same negative or positive association in relation to intention applies to intelligent design of plants. If we are convinced it is being done out of selfish, financial interests, we choose a term such as ‘genetic manipulation’. On the other hand, if we are convinced it is being done to be able to feed the world sustainably, we rather use terms such as ‘genetic modification’ or even ‘genetic enhancement’.
In any case, intelligent design of our mental models is happening, for good or for bad. In terms of education, it seems even necessary for our survival as a civilization. And although in one sense we are all subject to the imposition of mental models, by manipulation or education, I believe we also have the ability to reflect and to analyse these forces, and to make our own choices based on what we believe is the direction our individual and collective lives should take. I will get back to this important principle of choice later. But first, let’s look at the second concept, and move from adaptation to innovation.
Innovation
I proposed earlier that adaptation of an organism is a change in its internal state and its interaction with the environment, that genetic adaptation is heritable adaptation of a population, and a succession of genetic adaptations leads to evolution. But evolution, as it happened on this planet, seems to be more than just adaptation. It seems to be more than just an arms race. Moths that change their colour, plants that change their odour, fungi that become virulent on a new host – these changes appear to be just variations on a theme. It seems quite clear that much more dramatic changes have taken place. Sometimes, something emerged that appears to be really ‘new’. I will use the word innovation for this dramatic emergence of something new.
In biology, the word innovation is not as commonly used as adaptation. When we do speak of innovation at all, it is usually associated with rare, dramatic events that happened a long time ago, such as in the examples I mentioned earlier: the emergence of the ability to split water molecules using energy from light; or the ability to use the reverse reactions – between oxygen and carbohydrates – as a source of energy; or the emergence of land plants or land animals; or the emergence of the ability to fly in the animal kingdom, or the emergence of the ability to maintain a constant body temperature by mammals and birds; or the emergence of human language: these marvels we consider to be true innovations: these changes formed the starting point of a whole new development, they changed the world as we know it. But this is all hindsight – can we recognize a true innovation when it is happening?
In any case, using hindsight, evolutionary biologist Stephen Jay Gould has become famous with his concept of punctuated equilibrium, which means that evolution alternates between long periods of small incremental changes – equivalent, perhaps, to adaptation – and short periods of dramatic changes – equivalent, perhaps, to innovation[3]. Interestingly, the shorter periods of dramatic changes seem to have coincided with speciation: the splitting of an evolutionary lineage into many new lineages that, for a certain time, undergo relatively rapid changes, thus adding to the diversity of life. If the surviving sister lineage of a diversified lineage has undergone less dramatic speciation and change, we tend to call that lineage a ‘basal’ lineage, an ‘ancestral’ lineage, or even a ‘living fossil’, implying slower phenotypic change in the same amount of time. I think it will be interesting to see whether this correlation between speciation and innovation can be confirmed. This may not be so easy, since we know that extinctions have taken place and fossil evidence of extinct lineages is incomplete. Moreover, we would have to somehow quantify innovation.
But before considering the question of quantifying innovation, I briefly turn to the evolution of science. Here, I see a similarity of the concept of punctuated equilibrium of Stephen Gould to the theory of the well-known philosopher of science Thomas Kuhn. He famously suggested in 1962 that the scientific enterprise evolves through an alteration between relatively long periods of ‘normal science’ – periods of adaptation of existing theories or mental models – and shorter periods of ‘paradigm shifts’ –periods of innovation in mental models[4].
So, in both biology and science, it seems we use the word ‘innovation’ when we somehow perceive that something really new has been invented or has appeared. But, again, can we somehow objectify this newness? Can we go beyond an intuition of what is truly new? In science, it seems to me that we believe that we can and indeed we should go beyond intuition. After all, we judge research papers, grant proposals and patents on their degree of ‘novelty’ or ‘innovation’. Wouldn’t it be embarrassing if we would have to admit that this judgement is completely intuitive? Shouldn’t we measure it somehow? Objectively?
Of course, in one sense, nothing is ever new: it seems likely that the same atoms exist now as billions of years ago when the universe with its fixed set of elementary particles somehow came into being, the atom was born, and stars started to produce heavier elements from hydrogen. It also seems unlikely that new kinds of connection between these atoms will emerge. But the diversity of molecules that can be formed with this fixed set of atoms and connections between them is practically infinite, and the diversity of organisms that can be formed by the interplay between these molecules is infinite. So novelty is newness of form – the building blocks remain the same. But this, of course, just shifts the question of how to measure innovation to the question: How can we measure the ‘newness’ of a new combination of already existing elements?
When I look at the most obvious examples in biological evolution such as the ones mentioned before, to me innovation seems to be associated with the emergence of a new capability: the capability to split water, to use oxygen for energy, to breathe air, to regulate body temperature, to fly, or to speak. Over time, such a new capability has a transformative effect on the system, because new interactions become possible. So, there also seems to be the element of impact of the new capability that adds to the notion of innovation. Now, one problem with ‘measuring’ impact is that it takes time. The innovations that will change our future may already be among us, but can we confidently point them out? In the evolution of life on this planet, the next innovation may already be among us. Can we point it out?
In any case, let’s assume that innovation is indeed the emergence of a new capability – or a mental model – that has the potential to have a transforming impact on the system in which it emerged. We can now ask what kind of change in DNA or in our mental models, underlies this potential for transformative impact on the system? With regard to DNA, it seems to me that these changes tend to be more dramatic than the mutations that lead to adaptation. One example of a dramatic change in DNA is a burst of activation of a transposon – a parasitic genetic element that can multiply within the DNA of an organism and which can affect the activity of many genes in a single generation. A recently described example of this is the association of an expanded transposon family in vampire bats with genes that are likely involved in the ability of this unique creature to drink blood.[5] Another example is hybridization: a cross between two different but related species leading to offspring that combines traits of both parental species, something quite common in the plant kingdom. An even more dramatic example is the emergence of a new organism through symbiosis – an intimate and stable interaction between very different organisms that leads to new capabilities. Spectacular examples of symbioses include lichens, corals, mycorrhizae (which are fungi living in plant roots) and root nodules (which are nitrogen-fixing bacteria living in plant roots). The eukaryotic cell itself, with its unrivalled potential to form multicellular organisms including plants, animals and fungi, is a product of an ancient symbiosis on the cellular level.
In science and technology, innovation, again defined as a new capability or a new mental model within a society that has the potential to have a transforming impact on that society, also seems to result from new combinations – new combinations that lead to a leap forward in understanding or technology. This is essentially a creative process. Innovation requires the creative insight to make or recognize combinations that have not been seen before. As I mentioned before, the transformative impact of new combinations takes some time to become apparent. For example, the combination of genetics with biochemistry spawned the entire field of molecular biology, with the milestone discovery of the structure of the DNA molecule by Franklin, Watson and Crick in the 1950’s awarded a decade later with a Nobel Prize. Creative combinations labelled as innovation in science often take the form of applying insight or technology from one field to another. A recent spectacular example is the application of a newly discovered bacterial immune system to very specifically change genes in any organism without making them transgenic.[6]
So now, how can these ideas around the concept of innovation be applied to improve the health of the planet and advance human society?
It is, of course, generally recognized that to meet societal challenges, including in agriculture, energy and the preservation of the environment, technological innovation is essential. Innovation is indeed a key word for the financing of research projects by governments and granting agencies. To take just one example: in honour of the legacy of Thomas Kuhn, the philosopher of science I mentioned before, the American Chemical Society awards the “Thomas Kuhn Paradigm Shift Award" for the presentation of views that go beyond “mainstream scientific understanding”. Two criteria for this award are the “novelty of the viewpoint” and its “potential impact if it were to be widely accepted”[7].
Again, as I mentioned, innovation requires creativity. To innovate, therefore, we have to create the conditions in which a creative process can take place. This is a big question to explore; here I will only mention two conditions that I believe are required for innovation to take place: diversity and interaction. If true novelty arises from new combinations of diverse elements, then obviously the possibilities for innovation increase with the diversity of interacting elements, whether these are organisms, scientific disciplines or ideas. So a space or ecosystem with high diversity and a high degree of interaction seems to be required for nurturing innovation.
In biology, the relation between diversity, interaction and innovation is relevant for questions such as how sex evolved despite the more immediate benefit of clonal propagation, or how symbiosis evolved despite the danger of cheaters.
In the field of science, consider what would happen if researchers feel free to share and explore a diversity of ideas without fear of competition, without fearing negative consequences for their career or job security. Wouldn’t this stimulate collective, creative explorations?
Beyond biology, science and technology, I propose that the improvement of the health of the planet and of human society requires another type of innovation, namely innovation in our mental models, in our way of looking at the world. To take just one example, consider a mental model in which human beings are seen as fundamentally selfish creatures that only care about their own survival or at most the survival of their offspring or close kin. Suppose we would replace this mental model with the conviction that humans are spiritual beings that have a purpose – a purpose to develop virtues such as justice, generosity and compassion. And suppose we would extend that model to include the view that, together, human beings form a super-organism called humanity that has infinite capabilities for carrying forward an ever-advancing civilization. This mental model has, in fact, been introduced multiple times in human history in different languages and in different metaphors. Just consider what would happen if this mental model would be widely adopted. Would that not completely change the way we interact with each other? Would we not move from competition to mutual support, from fear to confidence, from self-interest to self-sacrifice, from indulgence in entertainment to a life of service?
Progress
This brings me to the third and most challenging concept, which is progress. At first sight, we could perhaps say that progress is simply the result of innovation. This seems to make sense when we look at biological evolution. The emergence of new capabilities such as photosynthesis, of focusing of light to create an image (as the eye does), of living on land, of flying, of keeping the body at a certain temperature, of human language, these innovations really seem to signify the progress of life on this planet. However, in contrast to innovation, the word progress and the related word advancement also have a moral dimension. With moral dimension I mean that these words attribute value to something. When we say that some organisms are more or advanced than others, or that certain human societies are more advanced than others, we attribute a value to organisms and societies. The more advanced organisms or societies are somehow more special, more worthy of preservation. But is that value attribution justified? Even if we were able to somehow measure the degree of innovation in evolution of organisms or societies, does ‘value’ necessarily follow from the ‘degree of innovation’? Are humans of more value than chimpansees because humans use language? Are mammals of more value than fish because they breathe air and can regulate their body temperature? Are animals of more value than plants? And can we really say that certain societies are of more value than others because they appear to show a higher degree of innovation and are therefore more advanced? Clearly, this is quite controversial.
To escape controversy, we may choose to retreat from this challenge and refrain from any value attribution. Perhaps we can take refuge in saying, for instance, that all animals, or even all life forms, are equally advanced – each in their own way – and are of equal value. The problem with this is that, in reality, we cannot really avoid attributing value. Take the importance that we attach to human rights. Few of us would deny the fundamental importance of human rights for our societies, and that these rights should apply equally to all humans on the planet because all are of ‘equal value’. But what of animal rights? Few would argue that animals should have the same rights as humans. But suppose we do say that, where do we draw the line? Large mammals only? Warm-blooded animals only? Vertebrates only? What about octopuses? Our moral values seem to be inextricably tide up with our notion that some life forms are more precious, more important to protect. Is it because they are really more advanced? Or are we just kidding ourselves and do we simply mean: they look more like us?
In biology, we try to be very disciplined and steer away from any moral judgement, all value attribution. I think that, in science, it is indeed important to do this, but it is not so easy. We automatically assign value. This is borne out by the language we use in our scientific discourse. Take, for example, the term ‘primitive’ or ‘ancestral’ when we talk about certain evolutionary lineages. To me it seems that, in biology, we find ourselves caught between the conflicting incentives to, on one hand, avoid value statements but, on the other, to somehow account for the innovation that has taken place in evolution. Perhaps a way out of this conflict is to use the word complexity to reflect the degree of innovation that has taken place in an evolutionary lineage. An increase in complexity is perhaps a more neutral, scientific word, compared to ‘progress’. But the challenge now becomes how to measure the complexity of an organism. Can we do that by counting the number of genes it has, or the number of levels of organisation, or the number of neurons or connections between neurons?
Interesting as this exercise is, I am convinced that we cannot and should not avoid the thorny issue of the assignment of value, because it has a much greater impact on ourselves and on our societies than the academic question of defining the degree of complexity of a system. We value progress. In fact, the experience of progress is perhaps the most important thing that motivates us. When we understand more of something, we feel great – we have made progress. When we can do something that we could not do before, we feel excited – we have made progress. When we move as a community from being divided to being united, we feel wonderful – we have made progress. In science, progress is everything. In our individual lives, progress is everything. It appears to me that defining progress only in terms of innovation, or of reaching a higher degree of complexity, does not do justice to our experience of progress. Progress is also the experience of a more coherent and unified understanding of the universe. Progress is also when we observe more beauty, when we see a desert transforming into a sea of flowers.
So now, how can these ideas around the concept of progress be applied to the betterment of society, or at least to the preservation of the planet? I think the main point I am trying to make is that we cannot avoid the moral dimension of life. We cannot isolate ourselves from the moral implications of our innovations, whether it concerns our technology or our mental models. They have a real impact on our planet and our societies. We constantly make choices based on what we value and we have to acknowledge that and not hide behind “I am just doing my job” or “I am just reporting the news” or “I am just a selfish collection of selfish genes”. We are morally bound to ask how our innovations should be applied. Do they serve to increase justice and beauty for all or rather the material interests of a few?
Taking responsibility for our choices can be challenging and maybe that is why we have a hard time doing that. If we seriously examine our moral framework, our purpose and our values on which we base our decisions, and really try to consistently apply our moral framework in every situation we encounter, the implications may be daunting. For instance, we may lose our job if we stick to integrity, or we may experience a significant reduction of material wealth if we stick to justice. It takes courage to take the full responsibility for the choices we make. I know this raises many questions such as: On what foundation do we build our moral framework? How do we build our moral framework? It is not easy to answer these questions, but they are vital questions if we want to make progress towards a more just civilization and a more beautiful planet.
Conclusions and Implications
Now, to finish, I will summarize my understanding of how we could apply what we have learned about adaptation, innovation and progress to science and society.
With regard to science, I propose that on the level of adaptation we need intelligence of an opportunistic flavour, of trying to be ‘smart’. For instance, as I suggested earlier, for our agriculture to become durable and remain productive, we need to adapt our crops in terms of genetics as well as in terms of our cropping systems, to keep all those viruses, bacteria and fungi under control, knowing that they will not go away and will keep adapting to changes in the environment and in our crops. We need to use our knowledge in a smart way, especially our knowledge of how the plant immune system works and how pathogens evade that immune system.
But we have to go beyond just being smart and also be innovative and therefore creative. This includes, for instance, the application of something that has been discovered in one field of research to other settings, such as the example I mentioned of the use of components of a bacterial immune system to edit genomes. Through curiosity-driven research we learn lessons from nature than can help us how to improve durability of our agriculture in new ways, such as the study of soil ecosystems to improve soil health. And beyond being smart and being creative, to achieve true progress I would suggest we also have to act with justice. For instance, we need to ask ourselves how innovations that improve agriculture can become accessible to all communities on this planet and not just benefit the revenue of a small number of countries or companies.
With regard to application of insights concerning adaptation, innovation and progress to society, my first conclusion is that there is hope for humanity. From biological evolution, we learn that life is not just an endless permutation of existing forms fuelled by relentless survival of the ‘fittest’ as defined by the existing system. Despite the struggle for survival, innovation happens: new and surprising combinations can emerge, leading to new capabilities with a transforming impact on the system. In the remarkably coherent system that we call a living cell, a great diversity of genes collaborate with each other and with an even greater diversity of proteins and other molecules. Symbiotic cells have emerged that over time have learned to differentiate and collaborate, resulting in the even more remarkable coherent systems that we call plants and animals. Even bad events such as the massive activation of transposons can lead to the emergence of surprising new capabilities. So I believe there is reason for hope, that humanity is able to evolve new capabilities through mutual support – new capabilities that will baffle our minds.
Hope creates the volition to act. And when we act we should be smart, creative and inclusive. We have to be smart to analyse the divisive forces that prevent progress and the unifying forces that support progress, and to identify what is needed for progress to occur. We need to learn to work in cycles of action, reflection and consultation. To innovate, we have to be creative, to combine previously unconnected processes to find innovative solutions to pressing problems – to creatively combine, for instance, physics and biology, education and research, religion and science. Creativity requires the creation of spaces of diversity and interaction, spaces of generous sharing and free exploration – in research as well as in education. To make real progress we need to be inclusive. Knowledge needs to be accessible to all. Education needs to serve the unfolding of the immense potential of all the inhabitants of this planet. Mutual support needs to be strengthened within and between all the diverse communities that make up our world community.
Finally, progress requires faith that real progress is possible, faith in the untapped potential of humankind, and faith that focussed effort can release that potential. I feel greatly encouraged by seeing the progress that has already been made, by nature, by humans, by the scientific community, and by humankind as a whole. However, to do even better, I believe we should ask ourselves the question: “Is there any greater blessing conceivable for a man [or woman], than that he [or she] should become the cause of the education, the development, the prosperity and honour of his fellow-creatures?” [8]
Gratitude
To end, a word of gratitude to all those who have created the conditions for me to be able to make progress, to the point where I stand here before you now, sharing a message of hope. In some or other way, you have all contributed to this. Instead of mentioning all your names, which are far too many, I will honour you by including you in groups of precious people. First, my parents – thank you for being my first educators. Second, my wife, son, daughter, brothers, sister, in-laws and other family members – thank you for walking the path of life with me. Third, my spiritual brothers and sisters – thank you for being my co-travellers in the path of service. Fourth, my colleagues and mentors in science, past and present – thank you for your indispensable support and inspiration. And finally, all gratitude is ultimately due, of course, to the One Whose Word instills new life into every human frame – thank you for making me conscious, for giving me a seeing eye and a hearing ear, and for patiently guiding me to Thy path.
References
[1] The essay was first published in American Biology Teacher in 1973.
[2] Drawn from conversations in programs of soul.com
[3] Eldredge, Niles and S. J. Gould (1972). "Punctuated equilibria: an alternative to phyletic gradualism" In T.J.M. Schopf, ed., Models in Paleobiology. San Francisco: Freeman Cooper. pp. 82-115.
[4] Thomas Samuel Kuhn (1962) The Structure of Scientific Revolutions (Chicago: The University of Chicago Press).
[5] Zepeda Mendoza et al. (2018) Nature Ecology & Evolution 2, p. 659–668.
[6] The CRISPR-Cas system (CRISPR: clustered regularly interspaced short palindromic repeats)
[7] http://www.acscomp.org/awards/thomas-kuhn-paradigm-shift-award
[8] Abdu’l-Bahá, in Secret of Divine Civilization
-
Right to education
Higher education for Bahá'ís in Iran
Since the 1980's Baha'is in Iran have been prevented from entering higher education – or expelled after enrollment.
-
Publications
- refereed (101)
- academic (12)
- professional (1)
- popular scientific (1)
- supervised theses (1)
- doctoral thesis (1)
- datasets (1)
2024
 Aalders, T. R., de Sain, M., Gawehns, F., Oudejans, N., Jak, Y. D., Dekker, H. L., Rep, M., van den Burg, H. A., & Takken, F. L. W. (2024). Specific members of the TOPLESS family are susceptibility genes for Fusarium wilt in tomato and Arabidopsis. Plant Biotechnology Journal, 22(1), 248-261. https://doi.org/10.1111/pbi.14183 [details]
Aalders, T. R., de Sain, M., Gawehns, F., Oudejans, N., Jak, Y. D., Dekker, H. L., Rep, M., van den Burg, H. A., & Takken, F. L. W. (2024). Specific members of the TOPLESS family are susceptibility genes for Fusarium wilt in tomato and Arabidopsis. Plant Biotechnology Journal, 22(1), 248-261. https://doi.org/10.1111/pbi.14183 [details]
2023
- Sabahi, F., Banihashemi, Z., Mirtalebi, M., Rep, M., & Cacciola, S. O. (2023). Molecular Variability of the Fusarium solani Species Complex Associated with Fusarium Wilt of Melon in Iran. Journal of Fungi, 9(4), Article 486. https://doi.org/10.3390/jof9040486
- Talbi, N., Fokkens, L., Audran, C., Petit-Houdenot, Y., Pouzet, C., Blaise, F., Gay, E. J., Rouxel, T., Balesdent, M. H., Rep, M., & Fudal, I. (2023). The neighbouring genes AvrLm10A and AvrLm10B are part of a large multigene family of cooperating effector genes conserved in Dothideomycetes and Sordariomycetes. Molecular Plant Pathology, 24(8), 914-931. https://doi.org/10.1111/mpp.13338
2021
 Ayukawa, Y., Asai, S., Gan, P., Tsushima, A., Ichihashi, Y., Shibata, A., Komatsu, K., Houterman, P. M., Rep, M., Shirasu, K., & Arie, T. (2021). A pair of effectors encoded on a conditionally dispensable chromosome of Fusarium oxysporum suppress host-specific immunity. Communications biology, 4, Article 707. https://doi.org/10.1038/s42003-021-02245-4 [details]
Ayukawa, Y., Asai, S., Gan, P., Tsushima, A., Ichihashi, Y., Shibata, A., Komatsu, K., Houterman, P. M., Rep, M., Shirasu, K., & Arie, T. (2021). A pair of effectors encoded on a conditionally dispensable chromosome of Fusarium oxysporum suppress host-specific immunity. Communications biology, 4, Article 707. https://doi.org/10.1038/s42003-021-02245-4 [details] Batson, A. M., Fokkens, L., Rep, M., & du Toit, L. J. (2021). Putative Effector Genes Distinguish Two Pathogenicity Groups of Fusarium oxysporum f. sp. spinaciae. Molecular Plant-Microbe Interactions, 34(2), 141-156. Advance online publication. https://doi.org/10.1094/MPMI-06-20-0145-R [details]
Batson, A. M., Fokkens, L., Rep, M., & du Toit, L. J. (2021). Putative Effector Genes Distinguish Two Pathogenicity Groups of Fusarium oxysporum f. sp. spinaciae. Molecular Plant-Microbe Interactions, 34(2), 141-156. Advance online publication. https://doi.org/10.1094/MPMI-06-20-0145-R [details] Constantin, M. E., Fokkens, L., de Sain, M., Takken, F. L. W., & Rep, M. (2021). Number of Candidate Effector Genes in Accessory Genomes Differentiates Pathogenic From Endophytic Fusarium oxysporum Strains. Frontiers in Plant Science, 12, Article 761740. https://doi.org/10.3389/fpls.2021.761740 [details]
Constantin, M. E., Fokkens, L., de Sain, M., Takken, F. L. W., & Rep, M. (2021). Number of Candidate Effector Genes in Accessory Genomes Differentiates Pathogenic From Endophytic Fusarium oxysporum Strains. Frontiers in Plant Science, 12, Article 761740. https://doi.org/10.3389/fpls.2021.761740 [details] Li, J., Fokkens, L., & Rep, M. (2021). A single gene in Fusarium oxysporum limits host range. Molecular Plant Pathology, 22(1), 108-116. https://doi.org/10.1111/mpp.13011 [details]
Li, J., Fokkens, L., & Rep, M. (2021). A single gene in Fusarium oxysporum limits host range. Molecular Plant Pathology, 22(1), 108-116. https://doi.org/10.1111/mpp.13011 [details] Sabahi, F., de Sain, M., Banihashemi, Z., & Rep, M. (2021). Comparative genomics of Fusarium oxysporum f. sp. melonis strains reveals nine lineages and a new sequence type of AvrFom2. Environmental Microbiology, 23(4), 2035. Advance online publication. https://doi.org/10.1111/1462-2920.15339 [details]
Sabahi, F., de Sain, M., Banihashemi, Z., & Rep, M. (2021). Comparative genomics of Fusarium oxysporum f. sp. melonis strains reveals nine lineages and a new sequence type of AvrFom2. Environmental Microbiology, 23(4), 2035. Advance online publication. https://doi.org/10.1111/1462-2920.15339 [details] de Lamo, F. J., Šimkovicová, M., Fresno, D. H., de Groot, T., Tintor, N., Rep, M., & Takken, F. L. W. (2021). Pattern-triggered immunity restricts host colonization by endophytic fusaria, but does not affect endophyte-mediated resistance. Molecular Plant Pathology, 22(2), 204-215. https://doi.org/10.1111/mpp.13018 [details]
de Lamo, F. J., Šimkovicová, M., Fresno, D. H., de Groot, T., Tintor, N., Rep, M., & Takken, F. L. W. (2021). Pattern-triggered immunity restricts host colonization by endophytic fusaria, but does not affect endophyte-mediated resistance. Molecular Plant Pathology, 22(2), 204-215. https://doi.org/10.1111/mpp.13018 [details]
2020
 Constantin, M. E., Vlieger, B. V., Takken, F. L. W., & Rep, M. (2020). Diminished Pathogen and Enhanced Endophyte Colonization upon CoInoculation of Endophytic and Pathogenic Fusarium Strains. Microorganisms, 8(4), Article 544. https://doi.org/10.3390/microorganisms8040544 [details]
Constantin, M. E., Vlieger, B. V., Takken, F. L. W., & Rep, M. (2020). Diminished Pathogen and Enhanced Endophyte Colonization upon CoInoculation of Endophytic and Pathogenic Fusarium Strains. Microorganisms, 8(4), Article 544. https://doi.org/10.3390/microorganisms8040544 [details] Constantin, M. E., de Lamo, F. J., Rep, M., & Takken, F. L. W. (2020). From laboratory to field: applying the Fo47 biocontrol strain in potato fields. European Journal of Plant Pathology, 158(3), 645–654. https://doi.org/10.1007/s10658-020-02106-6 [details]
Constantin, M. E., de Lamo, F. J., Rep, M., & Takken, F. L. W. (2020). From laboratory to field: applying the Fo47 biocontrol strain in potato fields. European Journal of Plant Pathology, 158(3), 645–654. https://doi.org/10.1007/s10658-020-02106-6 [details] Frantzeskakis, L., Di Pietro, A., Rep, M., Schirawski, J., Wu, C-H., & Panstruga, R. (2020). Rapid evolution in plant-microbe interactions - a molecular genomics perspective. New Phytologist, 225(3), 1134-1142. https://doi.org/10.1111/nph.15966 [details]
Frantzeskakis, L., Di Pietro, A., Rep, M., Schirawski, J., Wu, C-H., & Panstruga, R. (2020). Rapid evolution in plant-microbe interactions - a molecular genomics perspective. New Phytologist, 225(3), 1134-1142. https://doi.org/10.1111/nph.15966 [details] Li, J., Cornelissen, B., & Rep, M. (2020). Host-specificity factors in plant pathogenic fungi. Fungal Genetics and Biology, 144, Article 103447. Advance online publication. https://doi.org/10.1016/j.fgb.2020.103447 [details]
Li, J., Cornelissen, B., & Rep, M. (2020). Host-specificity factors in plant pathogenic fungi. Fungal Genetics and Biology, 144, Article 103447. Advance online publication. https://doi.org/10.1016/j.fgb.2020.103447 [details] Li, J., Fokkens, L., Conneely, L. J., & Rep, M. (2020). Partial pathogenicity chromosomes in Fusarium oxysporum are sufficient to cause disease and can be horizontally transferred. Environmental Microbiology, 22(12), 4985-5004. Advance online publication. https://doi.org/10.1111/1462-2920.15095 [details]
Li, J., Fokkens, L., Conneely, L. J., & Rep, M. (2020). Partial pathogenicity chromosomes in Fusarium oxysporum are sufficient to cause disease and can be horizontally transferred. Environmental Microbiology, 22(12), 4985-5004. Advance online publication. https://doi.org/10.1111/1462-2920.15095 [details] Li, J., Fokkens, L., van Dam, P., & Rep, M. (2020). Related mobile pathogenicity chromosomes in Fusarium oxysporum determine host range on cucurbits. Molecular Plant Pathology, 21(6), 761-776. Advance online publication. https://doi.org/10.1111/mpp.12927 [details]
Li, J., Fokkens, L., van Dam, P., & Rep, M. (2020). Related mobile pathogenicity chromosomes in Fusarium oxysporum determine host range on cucurbits. Molecular Plant Pathology, 21(6), 761-776. Advance online publication. https://doi.org/10.1111/mpp.12927 [details] Tintor, N., Paauw, M., Rep, M., & Takken, F. L. W. (2020). The root invading pathogen Fusarium oxysporum targets pattern-triggered immunity using both cytoplasmic and apoplastic effectors. New Phytologist, 227(5), 1479-1492. https://doi.org/10.1111/nph.16618 [details]
Tintor, N., Paauw, M., Rep, M., & Takken, F. L. W. (2020). The root invading pathogen Fusarium oxysporum targets pattern-triggered immunity using both cytoplasmic and apoplastic effectors. New Phytologist, 227(5), 1479-1492. https://doi.org/10.1111/nph.16618 [details] Zhang, Y., Yang, H., Turra, D., Zhou, S., Ayhan, D. H., DeIulio, G. A., Guo, L., Broz, K., Wiederhold, N., Coleman, J. J., Donnell, KO., Youngster, I., McAdam, A. J., Savinov, S., Shea, T., Young, S., Zeng, Q., Rep, M., Pearlman, E., ... Ma, L-J. (2020). The genome of opportunistic fungal pathogen Fusarium oxysporum carries a unique set of lineage-specific chromosomes. Communications biology, 3, Article 50. https://doi.org/10.1038/s42003-020-0770-2 [details]
Zhang, Y., Yang, H., Turra, D., Zhou, S., Ayhan, D. H., DeIulio, G. A., Guo, L., Broz, K., Wiederhold, N., Coleman, J. J., Donnell, KO., Youngster, I., McAdam, A. J., Savinov, S., Shea, T., Young, S., Zeng, Q., Rep, M., Pearlman, E., ... Ma, L-J. (2020). The genome of opportunistic fungal pathogen Fusarium oxysporum carries a unique set of lineage-specific chromosomes. Communications biology, 3, Article 50. https://doi.org/10.1038/s42003-020-0770-2 [details]
2019
 Constantin, M. E., de Lamo, F. J., Vlieger, B. V., Rep, M., & Takken, F. L. W. (2019). Endophyte-Mediated Resistance in Tomato to Fusarium oxysporum Is Independent of ET, JA, and SA. Frontiers in Plant Science, 10, Article 979. https://doi.org/10.3389/fpls.2019.00979 [details]
Constantin, M. E., de Lamo, F. J., Vlieger, B. V., Rep, M., & Takken, F. L. W. (2019). Endophyte-Mediated Resistance in Tomato to Fusarium oxysporum Is Independent of ET, JA, and SA. Frontiers in Plant Science, 10, Article 979. https://doi.org/10.3389/fpls.2019.00979 [details] Kesten, C., Gámez-Arjona, F. M., Menna, A., Scholl, S., Dora, S., Huerta, A. I., Huang, H-Y., Tintor, N., Kinoshita, T., Rep, M., Krebs, M., Schumacher, K., & Sánchez-Rodríguez, C. (2019). Pathogen-induced pH changes regulate the growth-defense balance in plants. EMBO Journal, 38(24), Article e101822. https://doi.org/10.15252/embj.2019101822 [details]
Kesten, C., Gámez-Arjona, F. M., Menna, A., Scholl, S., Dora, S., Huerta, A. I., Huang, H-Y., Tintor, N., Kinoshita, T., Rep, M., Krebs, M., Schumacher, K., & Sánchez-Rodríguez, C. (2019). Pathogen-induced pH changes regulate the growth-defense balance in plants. EMBO Journal, 38(24), Article e101822. https://doi.org/10.15252/embj.2019101822 [details]- de Lamo, F. J., Constantin, M. E., Fresno, D. H., Boeren, S., Rep, M., & Takken, F. L. W. (2019). Corrigendum: Xylem Sap Proteomics Reveals Distinct Differences Between R Gene- and Endophyte-Mediated Resistance Against Fusarium Wilt Disease in Tomato. Frontiers in Microbiology, 10. https://doi.org/10.3389/fmicb.2019.01872
2018
 de Lamo, F. J., Constantin, M. E., Fresno, D. H., Boeren, S., Rep, M., & Takken, F. L. W. (2018). Xylem Sap Proteomics Reveals Distinct Differences Between R Gene- and Endophyte-Mediated Resistance Against Fusarium Wilt Disease in Tomato. Frontiers in Microbiology, 9, Article 2977. https://doi.org/10.3389/fmicb.2018.02977 [details]
de Lamo, F. J., Constantin, M. E., Fresno, D. H., Boeren, S., Rep, M., & Takken, F. L. W. (2018). Xylem Sap Proteomics Reveals Distinct Differences Between R Gene- and Endophyte-Mediated Resistance Against Fusarium Wilt Disease in Tomato. Frontiers in Microbiology, 9, Article 2977. https://doi.org/10.3389/fmicb.2018.02977 [details] van Dam, P., de Sain, M., ter Horst, A., van der Gragt, M., & Rep, M. (2018). Use of Comparative Genomics-Based Markers for Discrimination of Host Specificity in Fusarium oxysporum. Applied and Environmental Microbiology, 84(1), Article e01868-17. https://doi.org/10.1128/AEM.01868-17 [details]
van Dam, P., de Sain, M., ter Horst, A., van der Gragt, M., & Rep, M. (2018). Use of Comparative Genomics-Based Markers for Discrimination of Host Specificity in Fusarium oxysporum. Applied and Environmental Microbiology, 84(1), Article e01868-17. https://doi.org/10.1128/AEM.01868-17 [details]
2017
 Brankovics, B., van Dam, P., Rep, M., de Hoog, G. S., van der Lee, T. A. J., Waalwijk, C., & van Diepeningen, A. D. (2017). Mitochondrial genomes reveal recombination in the presumed asexual Fusarium oxysporum species complex. BMC Genomics, 18, Article 735. https://doi.org/10.1186/s12864-017-4116-5 [details]
Brankovics, B., van Dam, P., Rep, M., de Hoog, G. S., van der Lee, T. A. J., Waalwijk, C., & van Diepeningen, A. D. (2017). Mitochondrial genomes reveal recombination in the presumed asexual Fusarium oxysporum species complex. BMC Genomics, 18, Article 735. https://doi.org/10.1186/s12864-017-4116-5 [details] Catanzariti, A-M., Do, H. T. T., Bru, P., de Sain, M., Thatcher, L. F., Rep, M., & Jones, D. A. (2017). The tomato I gene for Fusarium wilt resistance encodes an atypical leucine-rich repeat receptor-like protein whose function is nevertheless dependent on SOBIR1 and SERK3/BAK1. Plant Journal, 89(6), 1195–1209. Advance online publication. https://doi.org/10.1111/tpj.13458 [details]
Catanzariti, A-M., Do, H. T. T., Bru, P., de Sain, M., Thatcher, L. F., Rep, M., & Jones, D. A. (2017). The tomato I gene for Fusarium wilt resistance encodes an atypical leucine-rich repeat receptor-like protein whose function is nevertheless dependent on SOBIR1 and SERK3/BAK1. Plant Journal, 89(6), 1195–1209. Advance online publication. https://doi.org/10.1111/tpj.13458 [details] Chellappan Biju, V., Fokkens, L., Houterman, P. M., Rep, M., & Cornelissen, B. J. C. (2017). Multiple evolutionary trajectories have led to the emergence of races in Fusarium oxysporum f. sp. lycopersici. Applied and Environmental Microbiology, 83(4), Article e02548-16. Advance online publication. https://doi.org/10.1128/AEM.02548-16 [details]
Chellappan Biju, V., Fokkens, L., Houterman, P. M., Rep, M., & Cornelissen, B. J. C. (2017). Multiple evolutionary trajectories have led to the emergence of races in Fusarium oxysporum f. sp. lycopersici. Applied and Environmental Microbiology, 83(4), Article e02548-16. Advance online publication. https://doi.org/10.1128/AEM.02548-16 [details] Van der Does, D., Boutrot, F., Engelsdorf, T., Rhodes, J., McKenna, J. F., Vernhettes, S., Koevoets, I., Tintor, N., Veerabagu, M., Miedes, E., Segonzac, C., Roux, M., Breda, A. S., Hardtke, C. S., Molina, A., Rep, M., Testerink, C., Mouille, G., Höfte, H., ... Zipfel, C. (2017). The Arabidopsis leucine-rich repeat receptor kinase MIK2/LRR-KISS connects cell wall integrity sensing, root growth and response to abiotic and biotic stresses. PLOS Genetics, 13(6), Article e1006832. https://doi.org/10.1371/journal.pgen.1006832 [details]
Van der Does, D., Boutrot, F., Engelsdorf, T., Rhodes, J., McKenna, J. F., Vernhettes, S., Koevoets, I., Tintor, N., Veerabagu, M., Miedes, E., Segonzac, C., Roux, M., Breda, A. S., Hardtke, C. S., Molina, A., Rep, M., Testerink, C., Mouille, G., Höfte, H., ... Zipfel, C. (2017). The Arabidopsis leucine-rich repeat receptor kinase MIK2/LRR-KISS connects cell wall integrity sensing, root growth and response to abiotic and biotic stresses. PLOS Genetics, 13(6), Article e1006832. https://doi.org/10.1371/journal.pgen.1006832 [details] van Dam, P., & Rep, M. (2017). The Distribution of Miniature Impala Elements and SIX Genes in the Fusarium Genus is Suggestive of Horizontal Gene Transfer. Journal of molecular evolution, 85(1-2), 14-25. https://doi.org/10.1007/s00239-017-9801-0 [details]
van Dam, P., & Rep, M. (2017). The Distribution of Miniature Impala Elements and SIX Genes in the Fusarium Genus is Suggestive of Horizontal Gene Transfer. Journal of molecular evolution, 85(1-2), 14-25. https://doi.org/10.1007/s00239-017-9801-0 [details] van Dam, P., Fokkens, L., Ayukawa, Y., van der Gragt, M., Ter Horst, A., Brankovics, B., Houterman, P. M., Arie, T., & Rep, M. (2017). A mobile pathogenicity chromosome in Fusarium oxysporum for infection of multiple cucurbit species. Scientific Reports, 7, Article 9042. https://doi.org/10.1038/s41598-017-07995-y [details]
van Dam, P., Fokkens, L., Ayukawa, Y., van der Gragt, M., Ter Horst, A., Brankovics, B., Houterman, P. M., Arie, T., & Rep, M. (2017). A mobile pathogenicity chromosome in Fusarium oxysporum for infection of multiple cucurbit species. Scientific Reports, 7, Article 9042. https://doi.org/10.1038/s41598-017-07995-y [details] van der Does, H. C., & Rep, M. (2017). Adaptation to the Host Environment by Plant-Pathogenic Fungi. Annual Review of Phytopathology, 55, 427-450. https://doi.org/10.1146/annurev-phyto-080516-035551 [details]
van der Does, H. C., & Rep, M. (2017). Adaptation to the Host Environment by Plant-Pathogenic Fungi. Annual Review of Phytopathology, 55, 427-450. https://doi.org/10.1146/annurev-phyto-080516-035551 [details]
2016
 Chellapan, B. V., van Dam, P., Rep, M., Cornelissen, B. J. C., & Fokkens, L. (2016). Non-canonical Helitrons in Fusarium oxysporum. Mobile DNA, 7, Article 27. https://doi.org/10.1186/s13100-016-0083-7 [details]
Chellapan, B. V., van Dam, P., Rep, M., Cornelissen, B. J. C., & Fokkens, L. (2016). Non-canonical Helitrons in Fusarium oxysporum. Mobile DNA, 7, Article 27. https://doi.org/10.1186/s13100-016-0083-7 [details] Schmidt, S. M., Lukasiewicz, J., Farrer, R., van Dam, P., Bertoldo, C., & Rep, M. (2016). Comparative genomics of Fusarium oxysporum f. sp. melonis reveals the secreted protein recognized by the Fom-2 resistance gene in melon. New Phytologist, 209(1), 307-318. Advance online publication. https://doi.org/10.1111/nph.13584 [details]
Schmidt, S. M., Lukasiewicz, J., Farrer, R., van Dam, P., Bertoldo, C., & Rep, M. (2016). Comparative genomics of Fusarium oxysporum f. sp. melonis reveals the secreted protein recognized by the Fom-2 resistance gene in melon. New Phytologist, 209(1), 307-318. Advance online publication. https://doi.org/10.1111/nph.13584 [details] Shahi, S., Beerens, B., Bosch, M., Linmans, J., & Rep, M. (2016). Nuclear dynamics and genetic rearrangement in heterokaryotic colonies of Fusarium oxysporum. Fungal Genetics and Biology, 91, 20-31. Advance online publication. https://doi.org/10.1016/j.fgb.2016.03.003 [details]
Shahi, S., Beerens, B., Bosch, M., Linmans, J., & Rep, M. (2016). Nuclear dynamics and genetic rearrangement in heterokaryotic colonies of Fusarium oxysporum. Fungal Genetics and Biology, 91, 20-31. Advance online publication. https://doi.org/10.1016/j.fgb.2016.03.003 [details] Shahi, S., Fokkens, L., Houterman, P. M., & Rep, M. (2016). Suppressor of fusion, a Fusarium oxysporum homolog of Ndt80, is required for nutrient-dependent regulation of anastomosis. Fungal Genetics and Biology, 95, 49-57. Advance online publication. https://doi.org/10.1016/j.fgb.2016.08.005 [details]
Shahi, S., Fokkens, L., Houterman, P. M., & Rep, M. (2016). Suppressor of fusion, a Fusarium oxysporum homolog of Ndt80, is required for nutrient-dependent regulation of anastomosis. Fungal Genetics and Biology, 95, 49-57. Advance online publication. https://doi.org/10.1016/j.fgb.2016.08.005 [details] Vlaardingerbroek, I., Beerens, B., Rose, L., Fokkens, L., Cornelissen, B. J. C., & Rep, M. (2016). Exchange of core chromosomes and horizontal transfer of lineage-specific chromosomes in Fusarium oxysporum. Environmental Microbiology, 18(11), 3702-3713. Advance online publication. https://doi.org/10.1111/1462-2920.13281 [details]
Vlaardingerbroek, I., Beerens, B., Rose, L., Fokkens, L., Cornelissen, B. J. C., & Rep, M. (2016). Exchange of core chromosomes and horizontal transfer of lineage-specific chromosomes in Fusarium oxysporum. Environmental Microbiology, 18(11), 3702-3713. Advance online publication. https://doi.org/10.1111/1462-2920.13281 [details] Vlaardingerbroek, I., Beerens, B., Schmidt, S. M., Cornelissen, B. J. C., & Rep, M. (2016). Dispensable chromosomes in Fusarium oxysporum f.sp lycopersici. Molecular Plant Pathology, 17(9), 1455-1466. Advance online publication. https://doi.org/10.1111/mpp.12440 [details]
Vlaardingerbroek, I., Beerens, B., Schmidt, S. M., Cornelissen, B. J. C., & Rep, M. (2016). Dispensable chromosomes in Fusarium oxysporum f.sp lycopersici. Molecular Plant Pathology, 17(9), 1455-1466. Advance online publication. https://doi.org/10.1111/mpp.12440 [details] Zhang, L., Zhou, Z., Guo, Q., Fokkens, L., Miskei, M., Pócsi, I., Zhang, W., Chen, M., Wang, L., Sun, Y., Donzelli, B. G. G., Gibson, D. M., Nelson, D. R., Luo, J. G., Rep, M., Liu, H., Yang, S., Wang, J., Krasnoff, S. B., ... Lin, M. (2016). Insights into Adaptations to a Near-Obligate Nematode Endoparasitic Lifestyle from the Finished Genome of Drechmeria coniospora. Scientific Reports, 6, Article 23122. https://doi.org/10.1038/srep23122 [details]
Zhang, L., Zhou, Z., Guo, Q., Fokkens, L., Miskei, M., Pócsi, I., Zhang, W., Chen, M., Wang, L., Sun, Y., Donzelli, B. G. G., Gibson, D. M., Nelson, D. R., Luo, J. G., Rep, M., Liu, H., Yang, S., Wang, J., Krasnoff, S. B., ... Lin, M. (2016). Insights into Adaptations to a Near-Obligate Nematode Endoparasitic Lifestyle from the Finished Genome of Drechmeria coniospora. Scientific Reports, 6, Article 23122. https://doi.org/10.1038/srep23122 [details] van Dam, P., Fokkens, L., Schmidt, S. M., Linmans, J. J. J., Kistler, H. C., Ma, L-J., & Rep, M. (2016). Effector profiles distinguish formae speciales of Fusarium oxysporum. Environmental Microbiology, 18(11), 4087-4102. Advance online publication. https://doi.org/10.1111/1462-2920.13445 [details]
van Dam, P., Fokkens, L., Schmidt, S. M., Linmans, J. J. J., Kistler, H. C., Ma, L-J., & Rep, M. (2016). Effector profiles distinguish formae speciales of Fusarium oxysporum. Environmental Microbiology, 18(11), 4087-4102. Advance online publication. https://doi.org/10.1111/1462-2920.13445 [details] van der Does, H. C., Fokkens, L., Yang, A., Schmidt, S. M., Langereis, L., Lukasiewicz, J. M., Hughes, T. R., & Rep, M. (2016). Transcription Factors Encoded on Core and Accessory Chromosomes of Fusarium oxysporum Induce Expression of Effector Genes. PLOS Genetics, 12(11), Article e1006401. https://doi.org/10.1371/journal.pgen.1006401 [details]
van der Does, H. C., Fokkens, L., Yang, A., Schmidt, S. M., Langereis, L., Lukasiewicz, J. M., Hughes, T. R., & Rep, M. (2016). Transcription Factors Encoded on Core and Accessory Chromosomes of Fusarium oxysporum Induce Expression of Effector Genes. PLOS Genetics, 12(11), Article e1006401. https://doi.org/10.1371/journal.pgen.1006401 [details]
2015
 Gawehns, F., Ma, L., Bruning, O., Houterman, P. M., Boeren, S., Cornelissen, B. J. C., Rep, M., & Takken, F. L. W. (2015). The effector repertoire of Fusarium oxysporum determines the tomato xylem proteome composition following infection. Frontiers in Plant Science, 6, Article 967. https://doi.org/10.3389/fpls.2015.00967 [details]
Gawehns, F., Ma, L., Bruning, O., Houterman, P. M., Boeren, S., Cornelissen, B. J. C., Rep, M., & Takken, F. L. W. (2015). The effector repertoire of Fusarium oxysporum determines the tomato xylem proteome composition following infection. Frontiers in Plant Science, 6, Article 967. https://doi.org/10.3389/fpls.2015.00967 [details] Ma, L., Houterman, P. M., Gawehns, F., Cao, L., Sillo, F., Richter, H., Clavijo-Ortiz, M. J., Schmidt, S. M., Boeren, S., Vervoort, J., Cornelissen, B. J. C., Rep, M., & Takken, F. L. W. (2015). The AVR2-SIX5 gene pair is required to activate I-2-mediated immunity in tomato. New Phytologist, 208(2), 507-518. Advance online publication. https://doi.org/10.1111/nph.13455 [details]
Ma, L., Houterman, P. M., Gawehns, F., Cao, L., Sillo, F., Richter, H., Clavijo-Ortiz, M. J., Schmidt, S. M., Boeren, S., Vervoort, J., Cornelissen, B. J. C., Rep, M., & Takken, F. L. W. (2015). The AVR2-SIX5 gene pair is required to activate I-2-mediated immunity in tomato. New Phytologist, 208(2), 507-518. Advance online publication. https://doi.org/10.1111/nph.13455 [details] Shahi, S., Beerens, B., Manders, E. M. M., & Rep, M. (2015). Dynamics of the establishment of multinucleate compartments in Fusarium oxysporum. Eukaryotic Cell, 14(1), 78-85. https://doi.org/10.1128/EC.00200-14 [details]
Shahi, S., Beerens, B., Manders, E. M. M., & Rep, M. (2015). Dynamics of the establishment of multinucleate compartments in Fusarium oxysporum. Eukaryotic Cell, 14(1), 78-85. https://doi.org/10.1128/EC.00200-14 [details] Vlaardingerbroek, I., Beerens, B., Shahi, S., & Rep, M. (2015). Fluorescence Assisted Selection of Transformants (FAST): Using flow cytometry to select fungal transformants. Fungal Genetics and Biology, 76, 104-109. Advance online publication. https://doi.org/10.1016/j.fgb.2015.02.003 [details]
Vlaardingerbroek, I., Beerens, B., Shahi, S., & Rep, M. (2015). Fluorescence Assisted Selection of Transformants (FAST): Using flow cytometry to select fungal transformants. Fungal Genetics and Biology, 76, 104-109. Advance online publication. https://doi.org/10.1016/j.fgb.2015.02.003 [details] de Sain, M., & Rep, M. (2015). The Role of Pathogen-Secreted Proteins in Fungal Vascular Wilt Diseases. International Journal of Molecular Sciences, 16(10), 23970-23993. https://doi.org/10.3390/ijms161023970 [details]
de Sain, M., & Rep, M. (2015). The Role of Pathogen-Secreted Proteins in Fungal Vascular Wilt Diseases. International Journal of Molecular Sciences, 16(10), 23970-23993. https://doi.org/10.3390/ijms161023970 [details]
2014
- Gawehns, F., Houterman, P. M., Ait Ichou, F., Michielse, C. B., Hijdra, M., Cornelissen, B. J. C., Rep, M., & Takken, F. (2014). The Fusarium oxysporum effector Six6 contributes to virulence and suppresses I-2 mediated cell death. Molecular Plant-Microbe Interactions, 27(4), 336-348. https://doi.org/10.1094/MPMI-11-13-0330-R [details]
- Jonkers, W., Xayamongkhon, H., Haas, M., Olivain, C., van der Does, H. C., Broz, K., Rep, M., Alabouvette, C., Steinberg, C., & Kistler, H. C. (2014). EBR1 genomic expansion and its role in virulence of Fusarium species. Environmental Microbiology, 16(7), 1982-2003. https://doi.org/10.1111/1462-2920.12331 [details]
- Kang, S., Demers, J., Jimenez-Gasco, M. M., & Rep, M. (2014). Fusarium oxysporum. In R. A. Dean, A. Lichens-Park, & C. Kole (Eds.), Genomics of plant-associated fungi and oomycetes: dicot pathogens (pp. 99-119). Springer. https://doi.org/10.1007/978-3-662-44056-8_5 [details]
2013
- Kistler, H. C., Rep, M., & Ma, L-J. (2013). Structural dynamics of Fusarium genomes. In D. W. Brown, & R. H. Proctor (Eds.), Fusarium: genomics, molecular and cellular biology (pp. 31-41). Caister Academic Press. [details]
 Schmidt, S. M., Houterman, P. M., Schreiver, I., Ma, L., Amyotte, S., Chellappan, B., Boeren, S., Takken, F. L. W., & Rep, M. (2013). MITEs in the promoters of effector genes allow prediction of novel virulence genes in Fusarium oxysporum. BMC Genomics, 14, 119. https://doi.org/10.1186/1471-2164-14-119 [details]
Schmidt, S. M., Houterman, P. M., Schreiver, I., Ma, L., Amyotte, S., Chellappan, B., Boeren, S., Takken, F. L. W., & Rep, M. (2013). MITEs in the promoters of effector genes allow prediction of novel virulence genes in Fusarium oxysporum. BMC Genomics, 14, 119. https://doi.org/10.1186/1471-2164-14-119 [details]
2012
- Gonorazky, G., Laxalt, A. M., Dekker, H. L., Rep, M., Munnik, T., Testerink, C., & de la Canal, L. (2012). Phosphatidylinositol 4-phosphate is associated to extracellular lipoproteic fractions and is detected in tomato apoplastic fluid. Plant Biology, 14(1), 41-49. https://doi.org/10.1111/j.1438-8677.2011.00488.x [details]
- Lievens, B., Hanssen, I. M., & Rep, M. (2012). Recent Developments in the Detection and Identification of Formae Speciales and Races of Fusarium oxysporum: From Pathogenicity Testing to Molecular Diagnostics. In M. L. Gullino, J. Katan, & A. Garibaldi (Eds.), Fusarium Wilts of Greenhouse Vegetable and Ornamental Crops (pp. 47-55). American Phytopathological Society. [details]
- Ma, L-J., Kistler, H. C., & Rep, M. (2012). Evolution of Plant Pathogenicity in Fusarium Species. In L. D. Sibley, B. J. Howlett, & J. Heitman (Eds.), Evolution of Virulence in Eukaryotic Microbes (pp. 485-500). Wiley-Blackwell. [details]
- Michielse, C. B., Reijnen, L., Olivain, C., Alabouvette, C., & Rep, M. (2012). Degradation of aromatic compounds through the beta-ketoadipate pathway is required for pathogenicity of the tomato wilt pathogen Fusarium oxysporum f. sp. lycopersici. Molecular Plant Pathology, 13(9), 1089-1100. Advance online publication. https://doi.org/10.1111/j.1364-3703.2012.00818.x [details]
- Rodríguez-Ortiz, R., Michielse, C., Rep, M., Limón, M. C., & Avalos, J. (2012). Genetic basis of carotenoid overproduction in Fusarium oxysporum. Fungal Genetics and Biology, 49(9), 684-696. Advance online publication. https://doi.org/10.1016/j.fgb.2012.06.007 [details]
2011
- Chakrabarti, A., Rep, M., Wang, B., Ashton, A., Dodds, P., & Ellis, E. (2011). Variation in potential effector genes distinguishing Australian and non-Australian isolates of the cotton wilt pathogen Fusarium oxysporum f.sp. vasinfectum. Plant Pathology, 60(2), 232-243. https://doi.org/10.1111/j.1365-3059.2010.02363.x [details]
- Jonkers, W., van Kan, J. A. L., Tijm, P., Lee, Y-W., Tudzynski, P., Rep, M., & Michielse, C. B. (2011). The FRP1 F-box gene has different functions in sexuality, pathogenicity and metabolism in three fungal pathogens. Molecular Plant Pathology, 12(6), 548-563. https://doi.org/10.1111/j.1364-3703.2010.00689.x [details]
- Krasikov, V., Dekker, H. L., Rep, M., & Takken, F. L. W. (2011). The tomato xylem sap protein XSP10 is required for full susceptibility to Fusarium wilt disease. Journal of Experimental Botany, 62(3), 963-973. https://doi.org/10.1093/jxb/erq327 [details]
2010
- Ament, K., Krasikov, V., Allmann, S., Rep, M., Takken, F. L. W., & Schuurink, R. C. (2010). Methyl salicylate production in tomato affects biotic interactions. Plant Journal, 62(1), 124-134. https://doi.org/10.1111/j.1365-313X.2010.04132.x [details]
 Ma, L-J., van der Does, H. C., Borkovich, K. A., Coleman, J. J., Daboussi, M. J., Di Pietro, A., Dufresne, M., Freitag, M., Grabherr, M., Henrissat, B., Houterman, P. M., Kang, S., Shim, W. B., Woloshuk, C., Xie, X., Xu, J. -R., Antoniw, J., Baker, S. E., Bluhm, B. H., ... Rep, M. (2010). Comparative genomics reveals mobile pathogenicity chromosomes in Fusarium. Nature, 464(7287), 367-373. https://doi.org/10.1038/nature08850 [details]
Ma, L-J., van der Does, H. C., Borkovich, K. A., Coleman, J. J., Daboussi, M. J., Di Pietro, A., Dufresne, M., Freitag, M., Grabherr, M., Henrissat, B., Houterman, P. M., Kang, S., Shim, W. B., Woloshuk, C., Xie, X., Xu, J. -R., Antoniw, J., Baker, S. E., Bluhm, B. H., ... Rep, M. (2010). Comparative genomics reveals mobile pathogenicity chromosomes in Fusarium. Nature, 464(7287), 367-373. https://doi.org/10.1038/nature08850 [details]- Rep, M., & Kistler, H. C. (2010). The genomic organization of plant pathogenicity in Fusarium species. Current Opinion in Plant Biology, 13(4), 420-426. https://doi.org/10.1016/j.pbi.2010.04.004 [details]
- Takken, F., & Rep, M. (2010). The arms race between tomato and Fusarium oxysporum. Molecular Plant Pathology, 11(2), 309-314. https://doi.org/10.1111/j.1364-3703.2009.00605.x [details]
- Takken, F., van Ooijen, G., Lukasik, E., Ma, L., Gawehns, F., Houterman, P., & Rep, M. (2010). How to resist a tomato resistance gene? In H. Antoun, T. Avis, L. Brisson, D. Prévost, & M. Trepanier (Eds.), Proceedings of the 14th International Congress on Molecular Plant-Microbe Interactions: Quebec City, Quebec, Canada, July 19-23, 2009 (Biology of plant-microbe interactions; Vol. 7). International Society for Molecular Plant-Microbe Interactions. [details]
2009
 Coleman, J. J., Rounsley, S. D., Rodriguez-Carres, M., Kuo, A., Wasmann, C. C., Grimwood, J., Schmutz, J., Taga, M., White, G. J., Zhou, S., Schwartz, D. C., Freitag, M., Ma, L-J., Danchin, E. G. J., Henrissat, B., Coutinho, P. M., Nelson, D. R., Straney, D., Napoli, C. A., ... VanEtten, H. D. (2009). The genome of Nectria haematococca: Contribution of supernumerary chromosomes to gene expansion. PLOS Genetics, 5(8), e1000618. https://doi.org/10.1371/journal.pgen.1000618 [details]
Coleman, J. J., Rounsley, S. D., Rodriguez-Carres, M., Kuo, A., Wasmann, C. C., Grimwood, J., Schmutz, J., Taga, M., White, G. J., Zhou, S., Schwartz, D. C., Freitag, M., Ma, L-J., Danchin, E. G. J., Henrissat, B., Coutinho, P. M., Nelson, D. R., Straney, D., Napoli, C. A., ... VanEtten, H. D. (2009). The genome of Nectria haematococca: Contribution of supernumerary chromosomes to gene expansion. PLOS Genetics, 5(8), e1000618. https://doi.org/10.1371/journal.pgen.1000618 [details]- Houterman, P. M., Ma, L., van Ooijen, G., de Vroomen, M. J., Cornelissen, B. J. C., Takken, F. L. W., & Rep, M. (2009). The effector protein Avr2 of the xylem-colonizing fungus Fusarium oxysporum activates the tomato resistance protein I-2 intracellularly. Plant Journal, 58(6), 970-978. https://doi.org/10.1111/j.1365-313X.2009.03838.x [details]
- Jonkers, W., & Rep, M. (2009). Lessons from fungal F-box proteins. Eukaryotic Cell, 8(5), 677-695. https://doi.org/10.1128/EC.00386-08 [details]
- Jonkers, W., & Rep, M. (2009). Mutation of CRE1 in Fusarium oxysporum reverts the pathogenicity defects of the FRP1 deletion mutant. Molecular Microbiology, 74(5), 1100-1113. https://doi.org/10.1111/j.1365-2958.2009.06922.x [details]
- Jonkers, W., Andrade Rodrigues, C. D., & Rep, M. (2009). Impaired colonization and infection of tomato roots by the Δfrp1 mutant of Fusarium oxysporum correlates with reduced CWDE gene expression. Molecular Plant-Microbe Interactions, 22(5), 507-518. https://doi.org/10.1094/MPMI-22-5-0507 [details]
- Lievens, B., Houterman, P. M., & Rep, M. (2009). Effector gene screening allows unambiguous identification of Fusarium oxysporum f. sp. lycopersici races and discrimination from other formae speciales. FEMS Microbiology Letters, 300(2), 201-215. https://doi.org/10.1111/j.1574-6968.2009.01783.x [details]
- Lievens, B., van Baarlen, P., Verreth, C., Van Kerckhove, S., Rep, M., & Thomma, B. P. H. J. (2009). Evolutionary relationships between Fusarium oxysporum f. sp. lycopersici and F. oxysporum f. sp. radicis-lycopersici isolates inferred from mating type, elongation factor-1α and exopolygalacturonase sequences. Mycological Research, 113(10), 1181-1191. https://doi.org/10.1016/j.mycres.2009.07.019 [details]
- Michielse, C. B., & Rep, M. (2009). Pathogen profile update: Fusarium oxysporum. Molecular Plant Pathology, 10(3), 311-324. https://doi.org/10.1111/J.1364-3703.2009.00538.X [details]
 Michielse, C. B., van Wijk, R., Reijnen, L., Cornelissen, B. J. C., & Rep, M. (2009). Insight into the molecular requirements for pathogenicity of Fusarium oxysporum f. sp. lycopersici through large-scale insertional mutagenesis. Genome Biology, 10(1), R4. https://doi.org/10.1186/gb-2009-10-1-r4 [details]
Michielse, C. B., van Wijk, R., Reijnen, L., Cornelissen, B. J. C., & Rep, M. (2009). Insight into the molecular requirements for pathogenicity of Fusarium oxysporum f. sp. lycopersici through large-scale insertional mutagenesis. Genome Biology, 10(1), R4. https://doi.org/10.1186/gb-2009-10-1-r4 [details] Michielse, C. B., van Wijk, R., Reijnen, L., Manders, E. M. M., Boas, S., Olivain, C., Alabouvette, C., & Rep, M. (2009). The nuclear protein Sge1 of Fusarium oxysporum is required for parasitic growth. PLoS Pathogens, 5(10), e1000637. https://doi.org/10.1371/journal.ppat.1000637 [details]
Michielse, C. B., van Wijk, R., Reijnen, L., Manders, E. M. M., Boas, S., Olivain, C., Alabouvette, C., & Rep, M. (2009). The nuclear protein Sge1 of Fusarium oxysporum is required for parasitic growth. PLoS Pathogens, 5(10), e1000637. https://doi.org/10.1371/journal.ppat.1000637 [details]
2008
 Houterman, P. M., Cornelissen, B. J. C., & Rep, M. (2008). Suppression of plant resistance gene-based immunity by a fungal effector. PLoS Pathogens, 4(5), Article e1000061. https://doi.org/10.1371/journal.ppat.1000061 [details]
Houterman, P. M., Cornelissen, B. J. C., & Rep, M. (2008). Suppression of plant resistance gene-based immunity by a fungal effector. PLoS Pathogens, 4(5), Article e1000061. https://doi.org/10.1371/journal.ppat.1000061 [details]- Lievens, B., Rep, M., & Thomma, B. P. H. J. (2008). Recent developments in the molecular discrimination of formae speciales of Fusarium oxysporum. Pest Management Science, 64(8), 781-788. https://doi.org/10.1002/ps.1564 [details]
 van der Does, H. C., Duyvesteijn, R. G. E., Goltstein, P. M., van Schie, C. C. N., Manders, E. M. M., Cornelissen, B. J. C., & Rep, M. (2008). Expression of effector gene SIX1 of Fusarium oxysporum requires living plant cells. Fungal Genetics and Biology, 45(9), 1257-1264. https://doi.org/10.1016/j.fgb.2008.06.002 [details]
van der Does, H. C., Duyvesteijn, R. G. E., Goltstein, P. M., van Schie, C. C. N., Manders, E. M. M., Cornelissen, B. J. C., & Rep, M. (2008). Expression of effector gene SIX1 of Fusarium oxysporum requires living plant cells. Fungal Genetics and Biology, 45(9), 1257-1264. https://doi.org/10.1016/j.fgb.2008.06.002 [details]- van der Does, H. C., Lievens, B., Claes, L., Houterman, P. M., Cornelissen, B. J. C., & Rep, M. (2008). The presence of a virulence locus discriminates Fusarium oxysporum isolates causing tomato wilt from other isolates. Environmental Microbiology, 10(6), 1475-1485. https://doi.org/10.1111/j.1462-2920.2007.01561.x [details]
2007
- Cuomo, C. A., Güldener, U., Xu, J. -R., Trail, F., Turgeon, B. G., Di Pietro, A., Walton, J. D., Ma, L-J., Baker, S. E., Rep, M., Adam, G., Antoniw, J., Baldwin, T., Calvo, S., Chang, Y. ., DeCaprio, D., Gale, L. R., Gnerre, S., Goswami, R. S., ... Kistler, H. C. (2007). The Fusarium graminearum genome reveals a link between localized polymorphism and pathogen specialization. Science, 317(5843), 1400-1402. https://doi.org/10.1126/science.1143708 [details]
- Houterman, P. M., Speijer, D., Dekker, H. L., de Koster, C. G., Cornelissen, B. J. C., & Rep, M. (2007). The mixed proteome of Fusarium oxysporum-infected tomato xylem vessels. Molecular Plant Pathology, 8, 215-221. https://doi.org/10.1111/j.1364-3703.2007.00384.x [details]
- van der Does, H. C., & Rep, M. (2007). Virulence genes and the evolution of plant pathogenicity in fungi. Molecular Plant-Microbe Interactions, 20, 1175-1182. https://doi.org/10.1094/MPMI-20-10-1175 [details]
2006
- Kehr, J., & Rep, M. (2006). Protein extraction from xylem and phloem sap Plant Proteomics: Methods and Protocols. In V. Mechin, C. Damerval, M. Zivy, & H. Thiellement (Eds.), Methods in Molecular Biology (pp. 27-36). (355). Humana Press. [details]
- Rep, M., Duyvesteijn, R. G. E., Gale, L., Usgaard, T., Cornelissen, B. J. C., Ma, L-J., & Ward, T. J. (2006). The presence of GC-AG introns in N. crassa and other euascomycetes determined from analyses of complete genomes: implications for automated gene prediction. Genomics, 87, 338-347. https://doi.org/10.1016/j.ygeno.2005.11.014 [details]
- Rep, M., van der Does, H. C., Meijer, M., Houterman, P. M., Dekker, H. L., Speijer, D., de Koster, C. G., & Cornelissen, B. J. C. (2006). Secrets of xylem colonization- The xylem sap proteome of tomato infected with Fusarium oxysporum IS-MPM1. In C. Quinto, S. Lopes-Lara Sanchez, & O. Geiger (Eds.), Biology of Plant-Microbe Interactions. (pp. 597-601) [details]
- van Loon, L. C., Rep, M., & Pieterse, C. (2006). Significance of inducible defense-related proteins in infected plants. Annual Review of Phytopathology, 44, 7.1-7.28. https://doi.org/10.1146/annurev.phyto.44.070505.143321 [details]
2005
- Duyvesteijn, R. G. E., van Wijk, R., Boer, Y., Rep, M., Cornelissen, B. J. C., & Haring, M. A. (2005). Frp1 is a Fusarium oxysporum F-box protein required for pathogenicity on tomato. Molecular Microbiology, 57(4), 1051-1063. https://doi.org/10.1111/j.1365-2958.2005.04751.x [details]
- Leon-Kloosterziel, K. M., Verhagen, B. W. M., Keurentjes, J. J. B., Rep, M., van Loon, L. C., & Pieterse, C. M. J. (2005). Colonization of the Arabidopsis rhizosphere by fluorescent Pseudomonas spp. activates a root-specific, ethylene-responsive PR-5 gene in the vascular bundle. Plant Molecular Biology, 57, 731-748. https://doi.org/10.1007/s11103-005-3097-y [details]
- Rep, M. (2005). Small proteins of plant-pathogenic fungi secreted during host colonization. FEMS Microbiology Letters, 253, 19-27. https://doi.org/10.1016/j.femsle.2005.09.014 [details]
- Rep, M., Meijer, M., Houterman, P. M., van der Does, H. C., & Cornelissen, B. J. C. (2005). Fusarium oxysporum evades the 1-3-meidated resistance without altering the matching avirulence gene. Molecular Plant-Microbe Interactions, 18(1), 15-23. https://doi.org/10.1094/MPMI-18-0015 [details]
- Rep, M., van der Does, H. C., & Cornelissen, B. J. C. (2005). Drifter, a novel, low copy hAT-like transposon in Fusarium oxysporum is activated during starvation. Fungal Genetics and Biology, 42, 546-553. https://doi.org/10.1016/j.fgb.2005.03.007 [details]
2004
- Rep, M., van der Does, H. C., Meijer, M., van Wijk, R., Houterman, P. M., Dekker, H. L., de Koster, C. G., & Cornelissen, B. J. C. (2004). A small, cysteine-rich protein secreted by Fusarium oxysporum during colonization of xylem vessels is required for 1-3-mediated resistance in tomato. Molecular Microbiology, 53, 1373-1383. https://doi.org/10.1111/j.1365-2958.2004.04177.x [details]
2003
- Rep, M., Dekker, H. L., Vossen, J. H., de Boer, A. D., Houterman, P. M., de Koster, C. G., & Cornelissen, B. J. C. (2003). A tomato xylem sap protein represents a new family of small cysteine-rich proteins with structural similarity to lipid transfer proteins. FEBS Letters, 534, 82-86. https://doi.org/10.1016/S0014-5793(02)03788-2 [details]
- Teunissen, H. A. S., Rep, M., Houterman, P. M., Cornelissen, B. J. C., & Haring, M. A. (2003). Construction of a mitotic linkage map of Fusarium oxysporum based on Foxy-AFLPs. Molecular Genetics and Genomics, 269, 215-226. https://doi.org/10.1007/s00438-003-0825-3 [details]
2002
- Koerkamp, MG., Rep, M., Bussemaker, H. J., Hardy, G. P. M. A., Mul, A., Piekarska, K., Szigyarto, C. A., Teixeira De Mattos, M. J., & Tabak, H. F. (2002). Dissection of Transient Oxidative Stress Response in Saccharomyces cerevisiae by Using DNA Microarrays. Molecular Biology of the Cell, 13, 2783-2794. https://doi.org/10.1091/mbc.E02-02-0075 [details]
- Rep, M., Dekker, H. L., Vossen, J. H., de Boer, A., Houterman, P. M., Speijer, D., Back, J. W., de Koster, C. G., & Cornelissen, B. J. C. (2002). Mass spectrometric identification of isoforms of PR proteins in xylem sap of fungus-infected tomato. Plant Physiology, 130, 904-917. https://doi.org/10.1104/pp.007427 [details]
2001
- Rep, M., Proft, M., Remize, F., Tamás, M., Serrano, R., Thevelein, J. M., & Hohmann, S. (2001). The Saccharomyces cerevisiae Sko1p transcription factor mediates HOG pathway-dependent osmotic regulation of a set of genes encoding enzymes implicated in protection from oxidative damage. Molecular Microbiology, 40, 1067-1083. https://doi.org/10.1046/j.1365-2958.2001.02384.x [details]
2000
- Rep, M., Krantz, M., Thevelein, J. M., & Hohmann, S. (2000). The transcriptional response of Saccharomyces cerevisiae to osmotic shock. Hot1p and Msn2p/Msn4p are required for the induction of subsets of high osmolarity glycerol pathway-dependent genes. The Journal of Biological Chemistry, 275, 8290-8300. https://doi.org/10.1074/jbc.275.12.8290 [details]
- Tamas, M. J., Rep, M., Thevelein, J. M., & Hohmann, S. (2000). Stimulation of the yeast high osmolarity glycerol (HOG) pathway: evidence for a signal generated by a change in turgor rather than by water stress. FEBS Letters, 472, 159-165. https://doi.org/10.1016/S0014-5793(00)01445-9 [details]
1997
- Suzuki, C. K., Rep, M., van Dijl, J. M., Suda, K., Grivell, L. A., & Schatz, G. (1997). ATP-dependent proteases that also chaperone protein biogenesis. Trends in Biochemical Sciences, 22, 118-123. https://doi.org/10.1016/S0968-0004(97)01020-7 [details]
1996
 Guelin, EJ-M., Rep, M., & Grivell, L. A. (1996). Afg3p, a mitochondrial atp-dependent metalloprotease, is involved in degradation of mitochondrially-encoded cox1, cox3, cob, su6, su8 and su9 subunits of the inner membrane complex-III, complex-IV and complex-V. FEBS Letters, 381, 42-46. https://doi.org/10.1016/0014-5793(96)00074-9 [details]
Guelin, EJ-M., Rep, M., & Grivell, L. A. (1996). Afg3p, a mitochondrial atp-dependent metalloprotease, is involved in degradation of mitochondrially-encoded cox1, cox3, cob, su6, su8 and su9 subunits of the inner membrane complex-III, complex-IV and complex-V. FEBS Letters, 381, 42-46. https://doi.org/10.1016/0014-5793(96)00074-9 [details]- Rep, M., & Grivell, L. A. (1996). MBA1 encodes a mitochondrial membrane-associated protein required for biogenesis of the respiratory chain. FEBS Letters, 388, 185-188. https://doi.org/10.1016/0014-5793(96)00543-1 [details]
 Rep, M., & Grivell, L. A. (1996). The role of protein degradation in mitochondrial function and biogenesis. Current Genetics, 30, 267-380. [details]
Rep, M., & Grivell, L. A. (1996). The role of protein degradation in mitochondrial function and biogenesis. Current Genetics, 30, 267-380. [details] Rep, M., Nooy, J., Guelin, EJ-M., & Grivell, L. A. (1996). Three genes for mitochondrial proteins suppress null-mutations in both Afg3 and Rca1 when over-expressed. Current Genetics, 30, 206-211. https://doi.org/10.1007/s002940050122 [details]
Rep, M., Nooy, J., Guelin, EJ-M., & Grivell, L. A. (1996). Three genes for mitochondrial proteins suppress null-mutations in both Afg3 and Rca1 when over-expressed. Current Genetics, 30, 206-211. https://doi.org/10.1007/s002940050122 [details] Rep, M., van Dijl, J. M., Suda, K., Schatz, G., Grivell, L. A., & Suzuki, C. K. (1996). Promotion of mitochondrial membrane complex assembly by a proteiolytically inactive yeast lon. Science, 274, 103-106. https://doi.org/10.1126/science.274.5284.103 [details]
Rep, M., van Dijl, J. M., Suda, K., Schatz, G., Grivell, L. A., & Suzuki, C. K. (1996). Promotion of mitochondrial membrane complex assembly by a proteiolytically inactive yeast lon. Science, 274, 103-106. https://doi.org/10.1126/science.274.5284.103 [details]
2023
- Fokkens, L., & Rep, M. (2023). Population genomics reveals meiotic recombination in Fusarium oxysporum. Proceedings of the National Academy of Sciences of the United States of America, 120(30), Article e2309677120. https://doi.org/10.1073/pnas.2309677120
2019
 Franken, P., Takken, F. L. W., & Rep, M. (2019). Transcript accumulation in a trifold interaction gives insight into mechanisms of biocontrol. New Phytologist, 224(2), 547-549. https://doi.org/10.1111/nph.16141 [details]
Franken, P., Takken, F. L. W., & Rep, M. (2019). Transcript accumulation in a trifold interaction gives insight into mechanisms of biocontrol. New Phytologist, 224(2), 547-549. https://doi.org/10.1111/nph.16141 [details]
2012
- van der Does, H. C., & Rep, M. (2012). Horizontal transfer of supernumerary chromosomes in fungi. In M. D. Bolton, & B. P. H. J. Thomma (Eds.), Plant fungal pathogens: methods and protocols (pp. 427-437). (Methods in molecular biology; No. 835). Humana Press. https://doi.org/10.1007/978-1-61779-501-5_26 [details]
1995
- Grivell, L. A., Elzinga, S. D. J., Guelin, EJ-M., van Oosterum, K., Rep, M., & van der Spek, J. C. (1995). Nucleo-mitochondrial crosstalk in mitochondrial biogenesis. In Proc. 17th Int. Conf. Yeast Genetics and Molecular Biology (pp. S20-S20). [details]
- Guelin, EJ-M., Rep, M., & Grivell, L. A. (1995). Functional analysis of the yeast Afg3p, an ATP-dependent metalloprotease. In Proc. 1st Conf on the AAA family of ATPases (pp. 30-30). [details]
- Guelin, EJ-M., Rep, M., & Grivell, L. A. (1995). Role of the yeast Afg3p, an ATP-dependent metalloprotease, in mitochondrial biogenesis. In Proc. 1st Conf. on the AAA Family of ATPases (pp. 23-23). [details]
1994
- Grivell, L. A., Elzinga, S. D. J., van Oosterum, K., Guelin, EJ-M., Rep, M., & van der Spek, J. C. (1994). Novel features of mitochondrial gene expression in yeast. In 4th Spanish-Portuguese Congress of Biochemistry. (pp. 59-59). [details]
- Guelin, EJ-M., Rep, M., & Grivell, L. A. (1994). Sequence of the AFG3 gene encoding a new member of the FtsH/Yme1/Tma subfamily of the AAA-protein family. Yeast, 10, 1389-1394. https://doi.org/10.1002/yea.320101016 [details]
- Rep, M., Guelin, EJ-M., & Grivell, L. A. (1994). A new member of the AAA protein family, AFG3, is involved in the synthesis or assembly of mitochondrially encoded subunits of respiratory enzyme complexes. In Int. Meeting on Yeast Genetics and Mol. Biology. (pp. 74-74). [details]
1993
- Pel, H. J., Rep, M., Dubbink, H. J., & Grivell, L. A. (1993). Single-Point Mutations in Domain-Ii of the Yeast Mitochondrial Release Factor Mrf-1 Affect Ribosome Binding. Nucleic Acids Research, 21(23), 5308-5315. https://doi.org/10.1093/nar/21.23.5308 [details]
1992
- Pel, H. J., Maat, C., Rep, M., & Grivell, L. A. (1992). The yeast nuclear gene MRF1 encodes a mitochondrial peptide chain release factor and cures several mitochondrial RNA splicing defects. Nucleic Acids Research, 20(23), 6339-6346. https://doi.org/10.1093/nar/20.23.6339 [details]
- Pel, H. J., Rep, M., & Grivell, L. A. (1992). Sequence comparison of new prokaryotic and mitochondrial members of the polypeptide chain release factor family predicts a five-domain model for release factor structure. Nucleic Acids Research, 20(17), 4423-4428. https://doi.org/10.1093/nar/20.17.4423 [details]
2011
- Rep, M. (2011). Genen lenen. Intermediair. http://documents.plant.wur.nl/pri/mycosphaerella/genenlenen-intermediair2011.pdf
1996
- Rep, M. (1996). The Role of ATP-dependentProteases in Mitochondrial Biogenesis. Febodruk. [details]
2017
- Brankovics, B., van Dam, P., de Hoog, G. S., van der Lee, T. A. J., van Diepeningen, A. D., Rep, M. & Waalwijk, C. (2017). Additional file 1 of Mitochondrial genomes reveal recombination in the presumed asexual Fusarium oxysporum species complex. Figshare. https://doi.org/10.6084/m9.figshare.c.3882718_d1.v1
This list of publications is extracted from the UvA-Current Research Information System. Questions? Ask the library or the Pure staff of your faculty / institute. Log in to Pure to edit your publications. Log in to Personal Page Publication Selection tool to manage the visibility of your publications on this list. -
Ancillary activities
-
BIHE
Faculty (online teaching) -
Reflect Academy
Senior adviseur -
Emoena
Lid van Adviesraad -
Bahai community
To encourage and advise
-
BIHE