Research lines Cellular and Circuit Neuroscience
-
Synaptic plasticity & behaviour - Helmut Kessels
Synaptic Plasticity & Behaviour (research line Helmut Kessels)
Memory encoding occurs through the selective strengthening and weakening of synapses within a circuit of neurons. A dominant mechanism for this synaptic plasticity is changing the number of AMPA-type glutamate receptors at the receiving end of the synapse.
In our group we use our knowledge of AMPA-receptor plasticity as a tool to study adaptive behavior in mice. By monitoring AMPA-receptor trafficking we can identify synapses that undergo experience-dependent changes during memory formation. By manipulating AMPA-receptor function we can influence animal behavior.
The study of AMPA-receptor trafficking not only feeds our fundamental knowledge of cognitive function, but also may learn us more about cognitive dysfunction. Alzheimer’s disease patients have difficulties acquiring new memories. How synaptic plasticity is corrupted in mouse models for Alzheimer’s disease is studied in our group.
-
Healthy Longevity and Epilepsy – Erwin van Vliet and Gil Vantomme

Almost 65 million people suffer from epilepsy, making it one of the most common neurological diseases worldwide. About one third of all epilepsy patients cannot be adequately treated with currently available anti-seizure medication. Epilepsy is particularly relevant in the ageing population, where it often co-occurs with neurodegenerative disorders and can severely impact independence and quality of life.
One of the major challenges in epilepsy research is the identification of new therapeutic targets and the development of novel treatment strategies. Deep brain stimulation of the thalamus is an established treatment option for patients with drug‑resistant epilepsy, but the optimal stimulation target within the thalamus, one that maximizes seizure control, supports cognitive function, and minimizes side effects, remains to be identified. The ketogenic diet is another treatment option, and novel diets are under investigation to improve compliance and reduce side effects. The identification of new targets will facilitate the development of novel therapeutic approaches.
Moreover, there is an urgent need for reliable biomarkers that can predict whether epilepsy will develop, e.g. after brain trauma, severe brain insults or during aging. Biomarkers could substantially improve the management of people with epilepsy and could lead to prevention in the right person at the right time, thereby contributing to brain health and healthy aging.
In parallel, we investigate how mechanisms of epileptogenesis intersect with those driving Alzheimer’s disease and other age-related neurodegenerative disorders. We aim to understand shared pathways, such as network hyperexcitability, neuroinflammation and blood–brain barrier dysfunction, and to identify biomarkers that are informative for both epilepsy and Alzheimer’s disease. This integrated approach is intended to support earlier diagnosis and more targeted interventions that preserve cognitive function and promote healthy longevity.
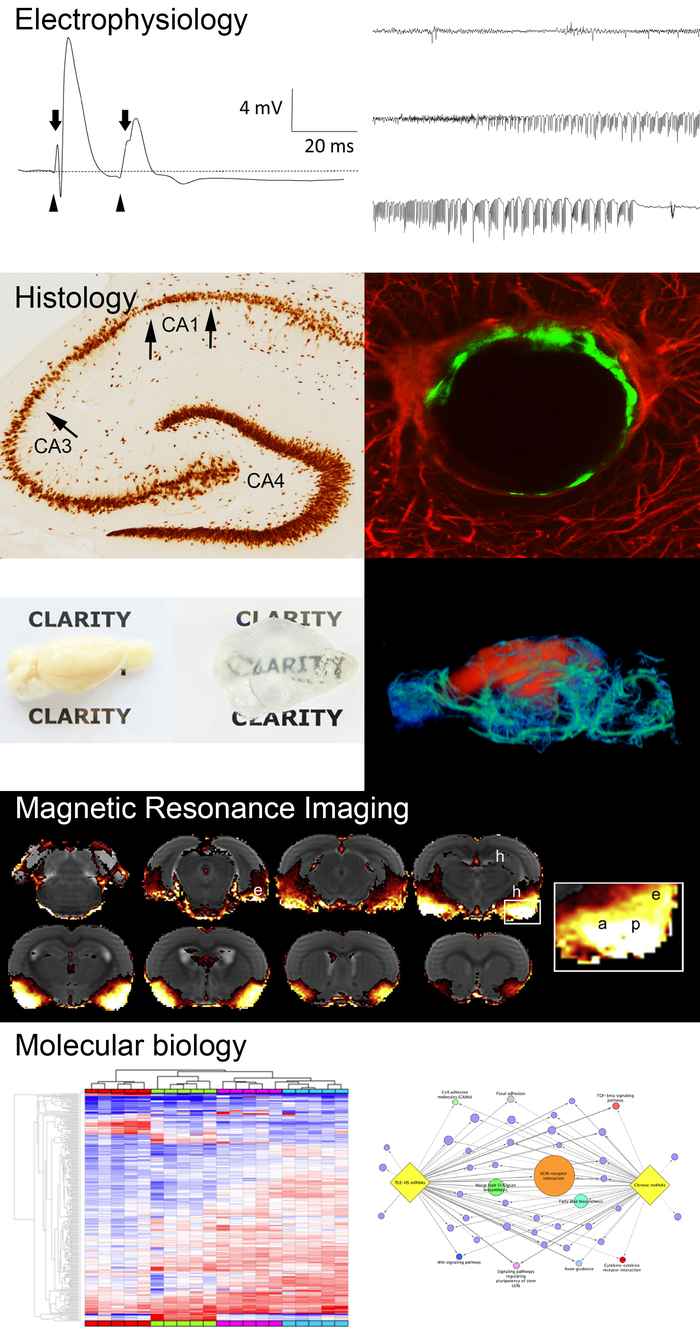
In our studies we use neurophysiological (EEG, field potentials in vivo and intracellular recordings in (cultured) brain slices), anatomical (tracing, immuno- and histochemistry), molecular (in situ hybridization, quantitative-PCR, western blot, microarrays, single cell- and nuclei sequencing, spatial transcriptomics) and imaging (magnetic resonance imaging) techniques to get insight in the processes that ultimately lead to epilepsy. Using in vitro and in vivo preclinical models and brain tissue from patients, we seek to unravel the mechanisms of drug-resistance and development of epilepsy (=epileptogenesis), to find biomarkers and to apply a treatment that could prevent or slow down epilepsy.
-
Endocannabinoid system – Pascal Chameau
Pascal Chameau investigates the endocannabinoid (eCB) system, a neuronal signaling system that comprises endogenous (lipid) ligands, synthesizing and metabolizing enzymes and cannabinoid receptors (CB1R and CB2R). It is involved in the mature brain, in modulating neuronal excitability and the transfer of information between neurons (via the inhibition of excitatory and inhibitory neurotransmitter release). In addition, the eCB system is a key regulatory signaling system of brain development. It is one of the first neuromodulator systems present in the developing brain and involved in neurogenesis, migration and specification of cortical neurons.
Our research focusses on understanding the role of the eCB system in cortical development and the consequences of exogenous cannabinoid exposure for cortical organization leading to cognitive deficits in adults. We aim at determining how the eCB system shapes cortical connectivity and excitability and how its alterations at early stages of cortical development affect cortical output and higher-order cognitive functions. In addition, we aim to determine whether alterations of the eCB systems can lead to neuronal migration defects which are associated with epilepsy.
In the mature brain, the eCB system is an interesting candidate for controlling (hyper)excitable neuronal (network) activity, for instance in cases of pathologies like epilepsy. Our previous research has shown that CB2R activation can reduce cellular excitability. We therefore also focus on the potential role of CB2 receptor activation in terminating pharmaco-resistant epileptic activity using both in vitro and in vivo electrophysiology in models for epilepsy.
-
Contribution of brain areas to cognition - Marlies Oostland
Marlies Oostland studies the contribution of the cerebellum and connected forebrain areas to cognition, using play as naturalistic behaviour to examine the development of neuronal dynamics for the formation of internal models and gating of sensory events. To do so, we monitor and perturb activity in cerebellar networks during playful behaviours, such as tickling and social play-fighting in rodents.