Research lines Molecular Biology and Microbial Food Safety
-
Food spoilage & pathogenic bacteria
Research line Prof. Stanley Brul
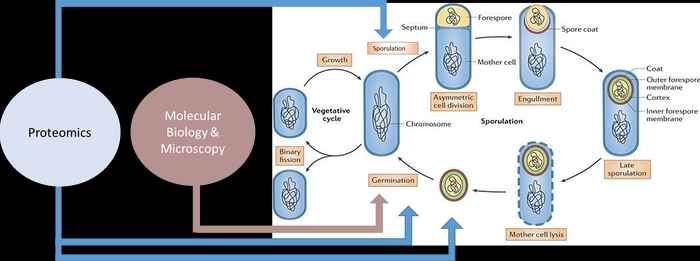
Spore forming organisms are spoilage organisms of prime-importance to the food industry due to their highly stress resistant endospores. These spores are also prominent in our gastro-intestinal tract. Their occurrence necessitates the application of harsh food preservation. The mechanistic basis of their extreme high thermal resistance (> 121˚C), as well as the molecular mechanisms of spore outgrowth are still characterized by many open questions.
With the help of molecular biology, advanced fluorescence microscopy and proteomics, we investigate ‘Germinosome’ complex required for spore germination, using the model system Bacillus subtilis and the pathogens Bacillus cereus and Peptoclostridium difficile. Studies are currently extended with the AUMC (location AMC) to the functional analysis of the gut microbiome.
-
Antibiotic resistance development in the food chain
Research line Prof. Benno ter Kuile
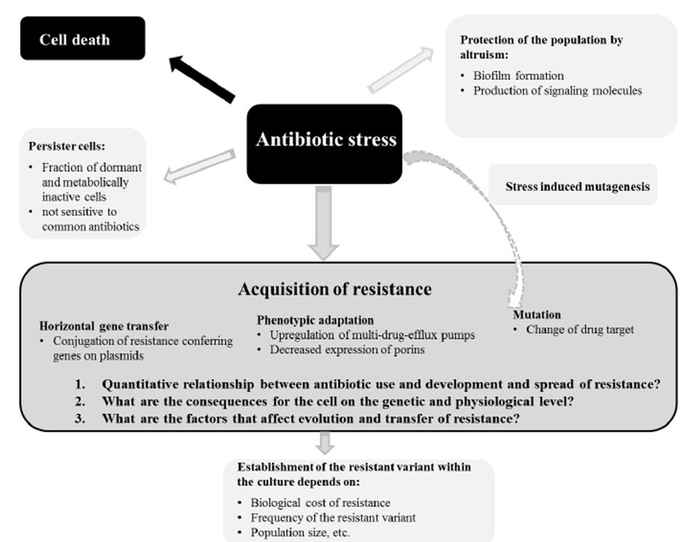
It is estimated that 50% to 80% of all antibiotics used are applied in agriculture. Veterinary and environmental microbes are often exposed to sublethal levels of antibiotics. Exposure to sublethal drug concentrations induces resistance, transfer of antimicrobial resistant (AMR) genes, and selection for already existing resistance. Novel approaches such as combination or alternating therapy are promising, but need to be investigated in detail before they can be implemented in daily practice.
-
How do species interactions affect evolution?
Research line Dr Meike Wortel
Micro-organisms live together with each other, and in the case of microbiota, also together with a host. Therefore, the evolution of micro-organisms is influenced by the interactions they have. Many interactions are mediated by external compounds, for example cross-feeding of metabolites. Using metabolic and mechanistic evolutionary models as well as laboratory (evolution) experiments with micro-organisms (from the gut microbiome) and nematode-bacteria interactions, the effects of species interactions on evolution are studied.
The main focus areas include: antibiotic resistance evolution in consortia of human gut microbes, co-evolution of C. elegans and E. coli and metabolic interactions in networks of human gut microbes and of C. elegans using bacteria as a food source. -
Fermentation for biobased production of (high-value) chemicals
Research line Prof. Jeroen Hugenholtz
As a crucial part of the developing Biobased Economy, the chemical industry is looking for alternative, sustainable, production processes for all/most of their bulk and fine chemicals, fuels, pharmaceuticals and food ingredients. Microbial fermentation is one of the key technologies for biobased production of these chemicals. The work, focusses on three areas of fermentation: (i) Anaerobic fermentation, involving Clostridium, for production of various alcohols from (biological) waste streams and C1-gasses (syngas), (ii) Lipid-producing and lipid-converting microorganisms such as Cryptococcus and Pseudomonas that can produce and convert various medium- and long-chain fatty acids, and (iii) Lactic acid bacteria as producer of natural flavors, of preservatives and for nutritional enrichment of food products. The described work is an important part of the Amsterdam Green Campus
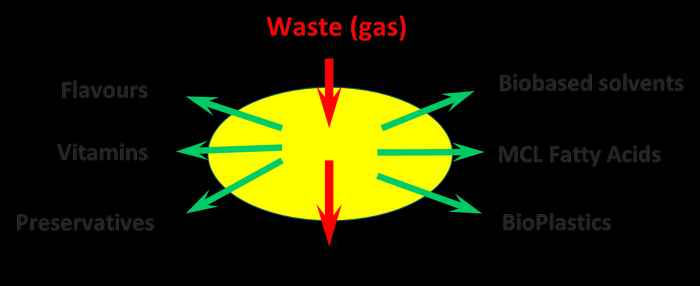
Biobased production of various useful chemicals 
Vegetable fermentation for increased shelf-life -
Microbiome-host interactions
Research line Dr Jianbo Zhang
We aim to study microbiome-host interactions and harness beneficial microbial functions to fight chronic and infectious diseases, with focus on (1) the development of microbiome and tissue engineering technologies; (2) elucidation of the mechanism of microbiome-host interactions in health and diseases; (3) engineering gut commensals and microbial consortia for smart therapeutic delivery.
You can also check the groups personal website.